Immunotherapy of non-Hodgkin's lymphoma with a defined ratio of CD8+ and CD4+ CD19-specific chimeric antigen receptor-modified T cells
- PMID: 27605551
- PMCID: PMC5045301
- DOI: 10.1126/scitranslmed.aaf8621
Immunotherapy of non-Hodgkin's lymphoma with a defined ratio of CD8+ and CD4+ CD19-specific chimeric antigen receptor-modified T cells
Abstract
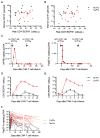
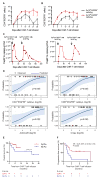
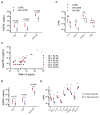
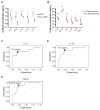
CD19-specific chimeric antigen receptor (CAR)-modified T cells have antitumor activity in B cell malignancies, but factors that affect toxicity and efficacy have been difficult to define because of differences in lymphodepletion and heterogeneity of CAR-T cells administered to individual patients. We conducted a clinical trial in which CD19 CAR-T cells were manufactured from defined T cell subsets and administered in a 1:1 CD4(+)/CD8(+) ratio of CAR-T cells to 32 adults with relapsed and/or refractory B cell non-Hodgkin's lymphoma after cyclophosphamide (Cy)-based lymphodepletion chemotherapy with or without fludarabine (Flu). Patients who received Cy/Flu lymphodepletion had increased CAR-T cell expansion and persistence, and higher response rates [50% complete remission (CR), 72% overall response rate (ORR)] than patients who received Cy-based lymphodepletion without Flu (8% CR, 50% ORR). The CR rate in patients treated with Cy/Flu at the maximally tolerated dose was 64% (82% ORR; n = 11). Cy/Flu minimized the effects of an immune response to the murine single-chain variable fragment component of the CAR, which limited CAR-T cell expansion and clinical efficacy in patients who received Cy-based lymphodepletion without Flu. Severe cytokine release syndrome (sCRS) and grade ≥3 neurotoxicity were observed in 13 and 28% of all patients, respectively. Serum biomarkers, one day after CAR-T cell infusion, correlated with subsequent sCRS and neurotoxicity. Immunotherapy with CD19 CAR-T cells in a defined CD4(+)/CD8(+) ratio allowed identification of correlative factors for CAR-T cell expansion, persistence, and toxicity, and facilitated optimization of lymphodepletion that improved disease response and overall and progression-free survival.
Copyright © 2016, American Association for the Advancement of Science.
Conflict of interest statement
CT and CB receive research funding from Juno Therapeutics and hold patents. DM receives research funding from Juno Therapeutics. SR receives research funding from and is a co-founder of Juno Therapeutics, and holds patents. MH holds a patent. DL is an employee of and has equity in Juno Therapeutics. FHCRC receives research funding from Juno Therapeutics.
Figures

References
-
- Turtle CJ, Hanafi LA, Berger C, Gooley TA, Cherian S, Hudecek M, Sommermeyer D, Melville K, Pender B, Budiarto TM, Robinson E, Steevens NN, Chaney C, Soma L, Chen X, Yeung C, Wood B, Li D, Cao J, Heimfeld S, Jensen MC, Riddell SR, Maloney DG. CD19 CAR-T cells of defined CD4+:CD8+ composition in adult B cell ALL patients. J Clin Invest. 2016;126:2123–2138. - PMC - PubMed
-
- Davila ML, Riviere I, Wang X, Bartido S, Park J, Curran K, Chung SS, Stefanski J, Borquez-Ojeda O, Olszewska M, Qu J, Wasielewska T, He Q, Fink M, Shinglot H, Youssif M, Satter M, Wang Y, Hosey J, Quintanilla H, Halton E, Bernal Y, Bouhassira DC, Arcila ME, Gonen M, Roboz GJ, Maslak P, Douer D, Frattini MG, Giralt S, Sadelain M, Brentjens R. Efficacy and toxicity management of 19–28z CAR T cell therapy in B cell acute lymphoblastic leukemia. Sci Transl Med. 2014;6:224ra25. - PMC - PubMed
-
- Brentjens RJ, Davila ML, Riviere I, Park J, Wang X, Cowell LG, Bartido S, Stefanski J, Taylor C, Olszewska M, Borquez-Ojeda O, Qu J, Wasielewska T, He Q, Bernal Y, Rijo IV, Hedvat C, Kobos R, Curran K, Steinherz P, Jurcic J, Rosenblat T, Maslak P, Frattini M, Sadelain M. CD19-targeted T cells rapidly induce molecular remissions in adults with chemotherapy-refractory acute lymphoblastic leukemia. Sci Transl Med. 2013;5:177ra38. - PMC - PubMed
-
- Lee DW, Kochenderfer JN, Stetler-Stevenson M, Cui YK, Delbrook C, Feldman SA, Fry TJ, Orentas R, Sabatino M, Shah NN, Steinberg SM, Stroncek D, Tschernia N, Yuan C, Zhang H, Zhang L, Rosenberg SA, Wayne AS, Mackall CL. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet. 2015;385:517–528. - PMC - PubMed
-
- Maude SL, Frey N, Shaw PA, Aplenc R, Barrett DM, Bunin NJ, Chew A, Gonzalez VE, Zheng Z, Lacey SF, Mahnke YD, Melenhorst JJ, Rheingold SR, Shen A, Teachey DT, Levine BL, June CH, Porter DL, Grupp SA. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med. 2014;371:1507–1517. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

