Chronic insulin treatment phosphorylates the renal Na-K-ATPase α1-subunit at serine 16/23 and reduces its activity involving PI3-kinase-dependent PKC activation
- PMID: 27605582
- PMCID: PMC5130455
- DOI: 10.1152/ajprenal.00355.2016
Chronic insulin treatment phosphorylates the renal Na-K-ATPase α1-subunit at serine 16/23 and reduces its activity involving PI3-kinase-dependent PKC activation
Abstract
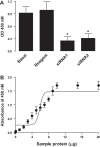
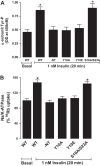
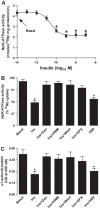
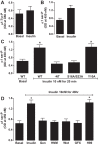
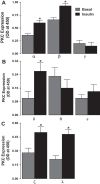
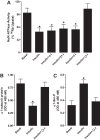
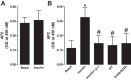
The regulation of Na-K-ATPase in various tissues is under the control of a number of hormones and peptides that exert both short- and long-term control over its activity. The present study was performed to investigate the effect of chronic insulin treatment on Na-K-ATPase in renal proximal tubular cells. Incubation of opossum kidney (OK) cells, transfected with the rat Na-K-ATPase α1-subunit, with 1 nmol/l insulin for 48 h decreased Na-K-ATPase activity. Insulin decreased α1-protein content and increased α1-serine phosphorylation and α1-adaptor protein 2 (AP2) interaction. Removal of the 26 NH2-terminal (-NT) amino acid from the α1-subunit containing serine/threonine sites abolished the insulin-mediated serine phosphorylation and inhibition of Na-K-ATPase. Substitution of serine 16 and 23 with alanine showed a comparable effect on -NT. Insulin increased the activity of protein kinase C (PKC), which was blocked by the phosphatidylinositol 3-kinase (PI3K) inhibitor wortmannin. Both PI3K and PKC inhibitors abolished the insulin-mediated inhibition of Na-K-ATPase. Insulin increased the expression of PKC-β1, -δ, -ξ, and-λ; however, only PKC-ξ/λ-specific inhibitors blocked insulin-induced phosphorylation and inhibition of Na-K-ATPase. Our data demonstrate that insulin activates the atypical PKC isoforms-ξ/λ via the PI3K pathway. PKC-ξ/λ-induced phosphorylation of the α1-subunit at serine 16 and 23 leads to AP2 recruitment, degradation, and a decrease in Na-K-ATPase activity.
Keywords: adaptor protein 2; kinase; serine kinase; serine phosphorylation; sodium transporter; tyrosine; tyrosine phosphorylation.
Copyright © 2016 the American Physiological Society.
Figures





References
-
- Al-Khalili L, Kotova O, Tsuchida H, Ehren I, Feraille E, Krook A, Chibalin AV. ERK1/2 mediates insulin stimulation of Na(+),K(+)-ATPase by phosphorylation of the alpha-subunit in human skeletal muscle cells. J Biol Chem 279: 25211–25218, 2004. - PubMed
-
- Banday AA, Siddiqui AH, Menezes MM, Hussain T. Insulin treatment enhances AT1 receptor function in OK cells. Am J Physiol Renal Physiol 288: F1213–F1219, 2005. - PubMed
-
- Bandyopadhyay G, Standaert ML, Galloway L, Moscat J, Farese RV. Evidence for involvement of protein kinase C (PKC)-zeta and noninvolvement of diacylglycerol-sensitive PKCs in insulin-stimulated glucose transport in L6 myotubes. Endocrinology 138: 4721–4731, 1997. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

