Hemorrhagic shock primes for lung vascular endothelial cell pyroptosis: role in pulmonary inflammation following LPS
- PMID: 27607578
- PMCID: PMC5059873
- DOI: 10.1038/cddis.2016.274
Hemorrhagic shock primes for lung vascular endothelial cell pyroptosis: role in pulmonary inflammation following LPS
Abstract
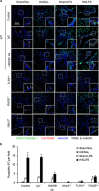
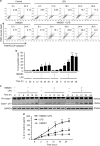
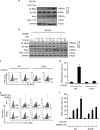
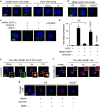
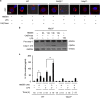
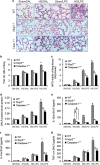
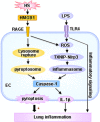
Hemorrhagic shock (HS) often renders patients more susceptible to lung injury by priming for an exaggerated response to a second infectious stimulus. Acute lung injury (ALI) is a major component of multiple organ dysfunction syndrome following HS and regularly serves as a major cause of patient mortality. The lung vascular endothelium is an active organ that has a central role in the development of ALI through synthesizing and releasing of a number of inflammatory mediators. Cell pyroptosis is a caspase-1-dependent regulated cell death, which features rapid plasma membrane rupture and release of proinflammatory intracellular contents. In this study, we demonstrated an important role of HS in priming for LPS-induced lung endothelial cell (EC) pyroptosis. We showed that LPS through TLR4 activates Nlrp3 (NACHT, LRR, and PYD domains containing protein 3) inflammasome in mouse lung vascular EC, and subsequently induces caspase-1 activation. However, HS induced release of high-mobility group box 1 (HMGB1), which acting through the receptor for advanced glycation end products initiates EC endocytosis of HMGB1, and subsequently triggers a cascade of molecular events, including cathepsin B release from ruptured lysosomes followed by pyroptosome formation and caspase-1 activation. These HS-induced events enhance LPS-induced EC pyroptosis. We further showed that lung vascular EC pyroptosis significantly exaggerates lung inflammation and injury. The present study explores a novel mechanism underlying HS-primed ALI and thus presents a potential therapeutic target for post-HS ALI.
Figures



Similar articles
-
TLR4-Upregulated IL-1β and IL-1RI Promote Alveolar Macrophage Pyroptosis and Lung Inflammation through an Autocrine Mechanism.Sci Rep. 2016 Aug 16;6:31663. doi: 10.1038/srep31663. Sci Rep. 2016. PMID: 27526865 Free PMC article.
-
Hemorrhagic shock augments Nlrp3 inflammasome activation in the lung through impaired pyrin induction.J Immunol. 2013 May 15;190(10):5247-55. doi: 10.4049/jimmunol.1203182. Epub 2013 Apr 12. J Immunol. 2013. PMID: 23585683 Free PMC article.
-
Hemorrhagic shock activation of NLRP3 inflammasome in lung endothelial cells.J Immunol. 2011 Nov 1;187(9):4809-17. doi: 10.4049/jimmunol.1102093. Epub 2011 Sep 21. J Immunol. 2011. PMID: 21940680 Free PMC article.
-
Mechanism and Regulation of NLRP3 Inflammasome Activation.Trends Biochem Sci. 2016 Dec;41(12):1012-1021. doi: 10.1016/j.tibs.2016.09.002. Epub 2016 Sep 23. Trends Biochem Sci. 2016. PMID: 27669650 Free PMC article. Review.
-
A potential new pathway for heparin treatment of sepsis-induced lung injury: inhibition of pulmonary endothelial cell pyroptosis by blocking hMGB1-LPS-induced caspase-11 activation.Front Cell Infect Microbiol. 2022 Sep 15;12:984835. doi: 10.3389/fcimb.2022.984835. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 36189354 Free PMC article. Review.
Cited by
-
Pyroptosis and respiratory diseases: A review of current knowledge.Front Immunol. 2022 Sep 30;13:920464. doi: 10.3389/fimmu.2022.920464. eCollection 2022. Front Immunol. 2022. PMID: 36248872 Free PMC article. Review.
-
Neutrophil extracellular traps promote macrophage pyroptosis in sepsis.Cell Death Dis. 2018 May 22;9(6):597. doi: 10.1038/s41419-018-0538-5. Cell Death Dis. 2018. PMID: 29789550 Free PMC article.
-
BCL6 attenuates renal inflammation via negative regulation of NLRP3 transcription.Cell Death Dis. 2017 Oct 26;8(10):e3156. doi: 10.1038/cddis.2017.567. Cell Death Dis. 2017. PMID: 29072703 Free PMC article.
-
The impact of neutrophil extracellular trap from patients with systemic lupus erythematosus on the viability, CD11b expression and oxidative burst of healthy neutrophils.BMC Immunol. 2021 Feb 5;22(1):12. doi: 10.1186/s12865-021-00402-2. BMC Immunol. 2021. PMID: 33546594 Free PMC article.
-
Non-apoptotic programmed cell deaths in diabetic pulmonary dysfunction: the new side of advanced glycation end products.Front Endocrinol (Lausanne). 2023 Oct 26;14:1126661. doi: 10.3389/fendo.2023.1126661. eCollection 2023. Front Endocrinol (Lausanne). 2023. PMID: 37964954 Free PMC article. Review.
References
-
- Rubenfeld GD, Caldwell E, Peabody E, Weaver J, Martin DP, Neff M et al. Incidence and outcomes of acute lung injury. N Engl J Med 2005; 353: 1685–1693. - PubMed
-
- Stapleton RD, Wang BM, Hudson LD, Rubenfeld GD, Caldwell ES, Steinberg KP. Causes and timing of death in patients with ARDS. Chest 2005; 128: 525–532. - PubMed
-
- Orfanos SE, Mavrommati I, Korovesi I, Roussos C. Pulmonary endothelium in acute lung injury: from basic science to the critically ill. Intensive Care Med 2004; 30: 1702–1714. - PubMed
-
- Matthay MA, Zimmerman GA, Esmon C, Bhattacharya J, Coller B, Doerschuk CM et al. Future research directions in acute lung injury: summary of a National Heart, Lung, and Blood Institute working group. Am J Respir Crit Care Med 2003; 167: 1027–1035. - PubMed
-
- Galluzzi L, Lopez-Soto A, Kumar S, Kroemer G. Caspases connect cell-death signaling to organismal homeostasis. Immunity 2016; 44: 221–231. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

