Model-based variables for the kinematic assessment of upper-extremity impairments in post-stroke patients
- PMID: 27609062
- PMCID: PMC5016877
- DOI: 10.1186/s12984-016-0187-9
Model-based variables for the kinematic assessment of upper-extremity impairments in post-stroke patients
Abstract
Background: Common scales for clinical evaluation of post-stroke upper-limb motor recovery are often complemented with kinematic parameters extracted from movement trajectories. However, there is no a general consensus on which parameters to use. Moreover, the selected variables may be redundant and highly correlated or, conversely, may incompletely sample the kinematic information from the trajectories. Here we sought to identify a set of clinically useful variables for an exhaustive but yet economical kinematic characterization of upper limb movements performed by post-stroke hemiparetic subjects.
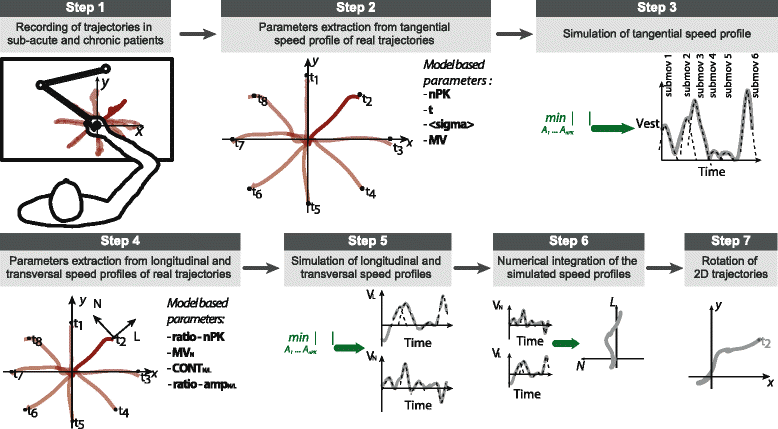
Methods: For this purpose, we pursued a top-down model-driven approach, seeking which kinematic parameters were pivotal for a computational model to generate trajectories of point-to-point planar movements similar to those made by post-stroke subjects at different levels of impairment.
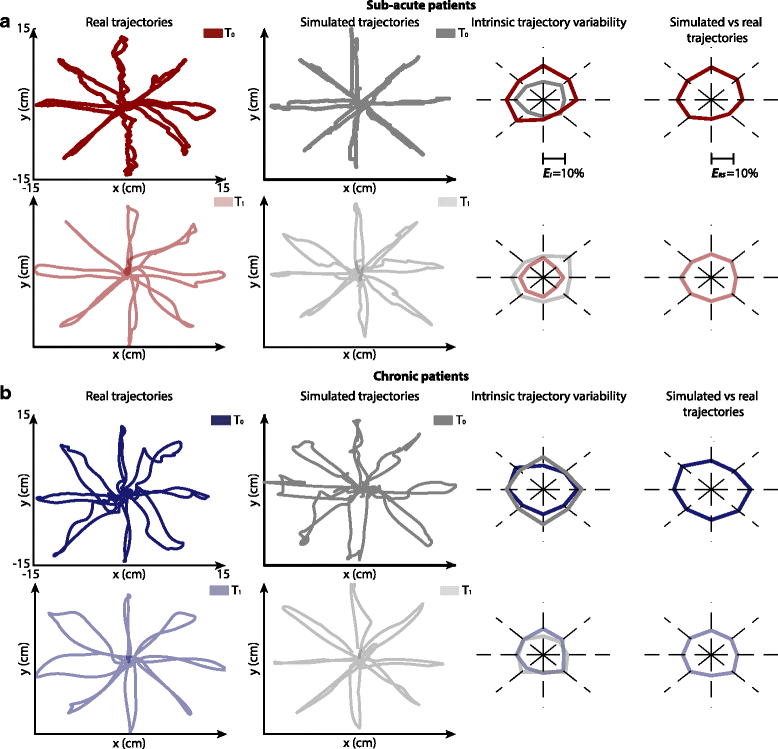
Results: The set of kinematic variables used in the model allowed for the generation of trajectories significantly similar to those of either sub-acute or chronic post-stroke patients at different time points during the therapy. Simulated trajectories also correctly reproduced many kinematic features of real movements, as assessed by an extensive set of kinematic metrics computed on both real and simulated curves. When inspected for redundancy, we found that variations in the variables used in the model were explained by three different underlying and unobserved factors related to movement efficiency, speed, and accuracy, possibly revealing different working mechanisms of recovery.
Conclusion: This study identified a set of measures capable of extensively characterizing the kinematics of upper limb movements performed by post-stroke subjects and of tracking changes of different motor improvement aspects throughout the rehabilitation process.
Keywords: Kinematics; Modeling; Robotic rehabilitation; Stroke.
Figures



References
-
- Gresham GE, Duncan PW, Stason WB. Post-stroke rehabilitation. Darby: DIANE Publishing; 1997.
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

