HIV-1 Tat Promotes Lysosomal Exocytosis in Astrocytes and Contributes to Astrocyte-mediated Tat Neurotoxicity
- PMID: 27609518
- PMCID: PMC5077215
- DOI: 10.1074/jbc.M116.731836
HIV-1 Tat Promotes Lysosomal Exocytosis in Astrocytes and Contributes to Astrocyte-mediated Tat Neurotoxicity
Abstract
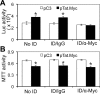
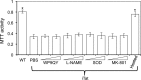
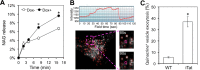
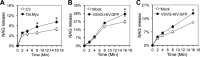
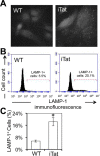
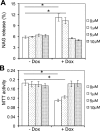
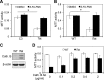
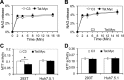
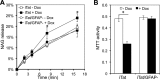
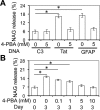
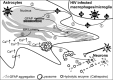
Tat interaction with astrocytes has been shown to be important for Tat neurotoxicity and HIV/neuroAIDS. We have recently shown that Tat expression leads to increased glial fibrillary acidic protein (GFAP) expression and aggregation and activation of unfolded protein response/endoplasmic reticulum (ER) stress in astrocytes and causes neurotoxicity. However, the exact molecular mechanism of astrocyte-mediated Tat neurotoxicity is not defined. In this study, we showed that neurotoxic factors other than Tat protein itself were present in the supernatant of Tat-expressing astrocytes. Two-dimensional gel electrophoresis and mass spectrometry revealed significantly elevated lysosomal hydrolytic enzymes and plasma membrane-associated proteins in the supernatant of Tat-expressing astrocytes. We confirmed that Tat expression and infection of pseudotyped HIV.GFP led to increased lysosomal exocytosis from mouse astrocytes and human astrocytes. We found that Tat-induced lysosomal exocytosis was tightly coupled to astrocyte-mediated Tat neurotoxicity. In addition, we demonstrated that Tat-induced lysosomal exocytosis was astrocyte-specific and required GFAP expression and was mediated by ER stress. Taken together, these results show for the first time that Tat promotes lysosomal exocytosis in astrocytes and causes neurotoxicity through GFAP activation and ER stress induction in astrocytes and suggest a common cascade through which aberrant astrocytosis/GFAP up-regulation potentiates neurotoxicity and contributes to neurodegenerative diseases.
Keywords: astrocyte; cathepsin B (CTSB); exocytosis; glial cell; human immunodeficiency virus (HIV); neuron.
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures
Similar articles
-
HIV-1 Tat Induces Unfolded Protein Response and Endoplasmic Reticulum Stress in Astrocytes and Causes Neurotoxicity through Glial Fibrillary Acidic Protein (GFAP) Activation and Aggregation.J Biol Chem. 2016 Oct 21;291(43):22819-22829. doi: 10.1074/jbc.M116.731828. Epub 2016 Sep 8. J Biol Chem. 2016. PMID: 27609520 Free PMC article.
-
Protection against human immunodeficiency virus type 1 Tat neurotoxicity by Ginkgo biloba extract EGb 761 involving glial fibrillary acidic protein.Am J Pathol. 2007 Dec;171(6):1923-35. doi: 10.2353/ajpath.2007.070333. Epub 2007 Nov 30. Am J Pathol. 2007. PMID: 18055541 Free PMC article.
-
Doxycycline-inducible and astrocyte-specific HIV-1 Tat transgenic mice (iTat) as an HIV/neuroAIDS model.J Neurovirol. 2018 Apr;24(2):168-179. doi: 10.1007/s13365-017-0598-9. Epub 2017 Nov 15. J Neurovirol. 2018. PMID: 29143286 Free PMC article. Review.
-
Astrocyte activation and dysfunction and neuron death by HIV-1 Tat expression in astrocytes.Mol Cell Neurosci. 2004 Nov;27(3):296-305. doi: 10.1016/j.mcn.2004.07.003. Mol Cell Neurosci. 2004. PMID: 15519244
-
Phosphatidylinositol (4,5)-bisphosphate-mediated pathophysiological effect of HIV-1 Tat protein.Biochimie. 2017 Oct;141:80-85. doi: 10.1016/j.biochi.2017.05.014. Epub 2017 May 23. Biochimie. 2017. PMID: 28549840 Review.
Cited by
-
HIV Nef Expression Down-modulated GFAP Expression and Altered Glutamate Uptake and Release and Proliferation in Astrocytes.Aging Dis. 2023 Feb 1;14(1):152-169. doi: 10.14336/AD.2022.0712. eCollection 2023 Feb 1. Aging Dis. 2023. PMID: 36818564 Free PMC article.
-
Disruption of the ADAM17/NF-κB feedback loop in astrocytes ameliorates HIV-1 Tat-induced inflammatory response and neuronal death.J Neurovirol. 2023 Jun;29(3):283-296. doi: 10.1007/s13365-023-01131-5. Epub 2023 Apr 25. J Neurovirol. 2023. PMID: 37185939
-
Functional impact of HIV-1 Tat on cells of the CNS and its role in HAND.Cell Mol Life Sci. 2020 Dec;77(24):5079-5099. doi: 10.1007/s00018-020-03561-4. Epub 2020 Jun 23. Cell Mol Life Sci. 2020. PMID: 32577796 Free PMC article. Review.
-
Impact of exosomal HIV-1 Tat expression on the human cellular proteome.Oncotarget. 2019 Sep 24;10(54):5632-5644. doi: 10.18632/oncotarget.27207. eCollection 2019 Sep 24. Oncotarget. 2019. PMID: 31608139 Free PMC article.
-
HIV, Tat and dopamine transmission.Neurobiol Dis. 2017 Sep;105:51-73. doi: 10.1016/j.nbd.2017.04.015. Epub 2017 Apr 27. Neurobiol Dis. 2017. PMID: 28457951 Free PMC article. Review.
References
-
- An S. F., Groves M., Gray F., and Scaravilli F. (1999) Early entry and widespread cellular involvement of HIV-1 DNA in brains of HIV-1 positive asymptomatic individuals. J. Neuropathol. Exp. Neurol. 58, 1156–1162 - PubMed
-
- Price R. W., and Brew B. J. (1988) The AIDS dementia complex. J. Infect. Dis. 158, 1079–1083 - PubMed
-
- Ellis R. J., Deutsch R., Heaton R. K., Marcotte T. D., McCutchan J. A., Nelson J. A., Abramson I., Thal L. J., Atkinson J. H., Wallace M. R., and Grant I. (1997) Neurocognitive impairment is an independent risk factor for death in HIV infection. San Diego HIV Neurobehavioral Research Center Group. Arch. Neurol. 54, 416–424 - PubMed
-
- Cohen R. A., and Gongvatana A. (2009) HIV-associated brain dysfunction in the era of HAART: reasons for hope, but continued concern. Neurology 73, 338–339 - PubMed
-
- Ellis R., Langford D., and Masliah E. (2007) HIV and antiretroviral therapy in the brain: neuronal injury and repair. Nat. Rev. Neurosci. 8, 33–44 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

