Multicentre, prospective, randomised, open-label, blinded end point trial of the efficacy of allopurinol therapy in improving cardiovascular outcomes in patients with ischaemic heart disease: protocol of the ALL-HEART study
- PMID: 27609859
- PMCID: PMC5020706
- DOI: 10.1136/bmjopen-2016-013774
Multicentre, prospective, randomised, open-label, blinded end point trial of the efficacy of allopurinol therapy in improving cardiovascular outcomes in patients with ischaemic heart disease: protocol of the ALL-HEART study
Abstract
Introduction: Ischaemic heart disease (IHD) is one of the most common causes of death in the UK and treatment of patients with IHD costs the National Health System (NHS) billions of pounds each year. Allopurinol is a xanthine oxidase inhibitor used to prevent gout that also has several positive effects on the cardiovascular system. The ALL-HEART study aims to determine whether allopurinol improves cardiovascular outcomes in patients with IHD.
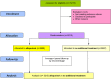
Methods and analysis: The ALL-HEART study is a multicentre, controlled, prospective, randomised, open-label blinded end point (PROBE) trial of allopurinol (up to 600 mg daily) versus no treatment in a 1:1 ratio, added to usual care, in 5215 patients aged 60 years and over with IHD. Patients are followed up by electronic record linkage and annual questionnaires for an average of 4 years. The primary outcome is the composite of non-fatal myocardial infarction, non-fatal stroke or cardiovascular death. Secondary outcomes include all-cause mortality, quality of life and cost-effectiveness of allopurinol. The study will end when 631 adjudicated primary outcomes have occurred. The study is powered at 80% to detect a 20% reduction in the primary end point for the intervention. Patient recruitment to the ALL-HEART study started in February 2014.
Ethics and dissemination: The study received ethical approval from the East of Scotland Research Ethics Service (EoSRES) REC 2 (13/ES/0104). The study is event-driven and results are expected after 2019. Results will be reported in peer-reviewed journals and at scientific meetings. Results will also be disseminated to guideline committees, NHS organisations and patient groups.
Trial registration number: 32017426, pre-results.
Keywords: allopurinol; cardiovascular outcomes; quality of life; uric acid.
Published by the BMJ Publishing Group Limited. For permission to use (where not already granted under a licence) please go to http://www.bmj.com/company/products-services/rights-and-licensing/
Figures
References
-
- Townsend N, Williams J, Bhatnagar P et al. Cardiovascular disease statistics 2014. London: British Heart Foundation, 2014.
-
- Spiekermann S, Landmesser U, Dikalov S et al. Electron spin resonance characterization of vascular xanthine and NAD(P)H oxidase activity in patients with coronary artery disease: relation to endothelium-dependent vasodilation. Circulation 2003;107:1383–9. 10.1161/01.CIR.0000056762.69302.46 - DOI - PubMed
-
- Doehner W, Schoene N, Rauchhaus M et al. Effects of xanthine oxidase inhibition with allopurinol on endothelial function and peripheral blood flow in hyperuricemic patients with chronic heart failure: results from 2 placebo-controlled studies. Circulation 2002;105:2619–24. 10.1161/01.CIR.0000017502.58595.ED - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources