Alcohol, Aldehyde, and Ketone Liberation and Intracellular Cargo Release through Peroxide-Mediated α-Boryl Ether Fragmentation
- PMID: 27636404
- PMCID: PMC7075644
- DOI: 10.1021/jacs.6b07890
Alcohol, Aldehyde, and Ketone Liberation and Intracellular Cargo Release through Peroxide-Mediated α-Boryl Ether Fragmentation
Abstract
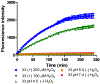
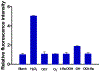
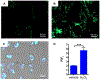
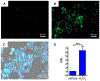
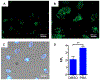
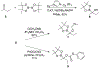
α-Boryl ethers, carbonates, and acetals, readily prepared from the corresponding alcohols that are accessed through ketone diboration, react rapidly with hydrogen peroxide to release alcohols, aldehydes, and ketones through the collapse of hemiacetal intermediates. Experiments with α-boryl acetals containing a latent fluorophore clearly demonstrate that cargo can be released inside cells in the presence of exogenous or endogenous hydrogen peroxide. These experiments show that this protocol can be used for drug activation in an oxidative environment without generating toxic byproducts.
Conflict of interest statement
The authors declare the following competing financial interest(s): The authors have applied for a provisional patent covering the drug release applications of this work.
Figures

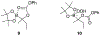
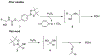

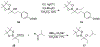
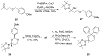


Similar articles
-
Air- and moisture-stable amphoteric molecules: enabling reagents in synthesis.Acc Chem Res. 2014 Apr 15;47(4):1029-40. doi: 10.1021/ar400210c. Epub 2014 Feb 4. Acc Chem Res. 2014. PMID: 24495255
-
Trifluoromethylation of ketones and aldehydes with Bu₃SnCF₃.J Org Chem. 2013 Aug 2;78(15):7749-53. doi: 10.1021/jo401099e. Epub 2013 Jul 16. J Org Chem. 2013. PMID: 23834264
-
1,5-Asymmetric induction in boron-mediated aldol reactions of beta-alkoxy methylketones.J Org Chem. 2008 Aug 15;73(16):6299-311. doi: 10.1021/jo8009165. Epub 2008 Jul 11. J Org Chem. 2008. PMID: 18616322
-
Transition metal catalysed reactions of alcohols using borrowing hydrogen methodology.Dalton Trans. 2009 Feb 7;(5):753-62. doi: 10.1039/b813383b. Epub 2008 Nov 21. Dalton Trans. 2009. PMID: 19156265 Review.
-
Enantioselective aldol reactions catalyzed by chiral phosphine oxides.Chem Rec. 2013 Aug;13(4):362-70. doi: 10.1002/tcr.201300004. Epub 2013 Jul 4. Chem Rec. 2013. PMID: 23828817 Review.
Cited by
-
Organelle-targeting ratiometric fluorescent probes: design principles, detection mechanisms, bio-applications, and challenges.Chem Sci. 2023 May 12;14(22):5842-5871. doi: 10.1039/d3sc01036h. eCollection 2023 Jun 7. Chem Sci. 2023. PMID: 37293660 Free PMC article. Review.
-
Difunctional oxidatively cleavable alkenyl boronates: application to cellular peroxide sensing from a fluorophore-quencher pair.Chem Commun (Camb). 2025 Feb 18;61(16):3375-3378. doi: 10.1039/d5cc00090d. Chem Commun (Camb). 2025. PMID: 39887215 Free PMC article.
-
MACA Fast and Efficient Method for Detecting H2O2 by a Dual-Locked Model Chemosensor.ACS Omega. 2021 May 28;6(23):14819-14823. doi: 10.1021/acsomega.1c00384. eCollection 2021 Jun 15. ACS Omega. 2021. PMID: 34151063 Free PMC article.
-
Potent and Selective Oxidatively Labile Ether-Based Prodrugs through Late-Stage Boronate Incorporation.Angew Chem Int Ed Engl. 2024 Oct 1;63(40):e202409229. doi: 10.1002/anie.202409229. Epub 2024 Sep 2. Angew Chem Int Ed Engl. 2024. PMID: 38986017
-
Organoboronic acids/esters as effective drug and prodrug candidates in cancer treatments: challenge and hope.J Enzyme Inhib Med Chem. 2023 Dec;38(1):2220084. doi: 10.1080/14756366.2023.2220084. J Enzyme Inhib Med Chem. 2023. PMID: 37318308 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

