Cathepsin K Deficiency Prevents the Aggravated Vascular Remodeling Response to Flow Cessation in ApoE-/- Mice
- PMID: 27636705
- PMCID: PMC5026377
- DOI: 10.1371/journal.pone.0162595
Cathepsin K Deficiency Prevents the Aggravated Vascular Remodeling Response to Flow Cessation in ApoE-/- Mice
Abstract
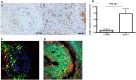
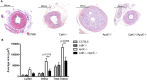
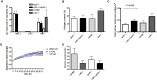
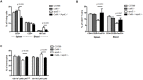
Cathepsin K (catK) is a potent lysosomal cysteine protease involved in extracellular matrix (ECM) degradation and inflammatory remodeling responses. Here we have investigated the contribution of catK deficiency on carotid arterial remodeling in response to flow cessation in apoE-/- and wild type (wt) background. Ligation-induced hyperplasia is considerably aggravated in apoE-/- versus wt mice. CatK protein expression was significantly increased in neointimal lesions of apoE-/- compared with wt mice, suggesting a role for catK in intimal hyperplasia under hyperlipidemic conditions. Surprisingly, CatK deficiency completely blunted the augmented hyperplastic response to flow cessation in apoE-/-, whereas vascular remodeling in wt mice was unaffected. As catK deficiency did neither alter lesion collagen content and elastic laminae fragmentation in vivo, we focused on effects of catK on (systemic) inflammatory responses. CatK deficiency significantly reduced circulating CD3 T-cell numbers, but increased the regulatory T cell subset in apoE-/- but not wt mice. Moreover, catK deficiency changed CD11b+Ly6G-Ly6C high monocyte distribution in apoE-/- but not wt mice and tended to favour macrophage M2a polarization. In conclusion, catK deficiency almost completely blunted the increased vascular remodeling response of apoE-/- mice to flow cessation, possibly by correcting hyperlipidemia-associated pro-inflammatory effects on the peripheral immune response.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

References
-
- Bossard MJ, Tomaszek TA, Thompson SK, Amegadzie BY, Hanning CR, Jones C, et al. Proteolytic activity of human osteoclast cathepsin K expression, purification, activation, and substrate identification. Journal of Biological Chemistry. 1996;271(21):12517–24. - PubMed
-
- Lutgens SP, Kisters N, Lutgens E, van Haaften RI, Evelo CT, de Winther MP, et al. Gene profiling of cathepsin K deficiency in atherogenesis: profibrotic but lipogenic. The Journal of pathology. 2006;210(3):334–43. - PubMed
-
- Lutgens E, Lutgens S, Faber B, Heeneman S, Gijbels M, de Winther M, et al. Disruption of the cathepsin K gene reduces atherosclerosis progression and induces plaque fibrosis but accelerates macrophage foam cell formation. Circulation. 2006;113(1):98–107. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

