Endoplasmic Reticulum Stress Links Oxidative Stress to Impaired Pancreatic Beta-Cell Function Caused by Human Oxidized LDL
- PMID: 27636901
- PMCID: PMC5026355
- DOI: 10.1371/journal.pone.0163046
Endoplasmic Reticulum Stress Links Oxidative Stress to Impaired Pancreatic Beta-Cell Function Caused by Human Oxidized LDL
Abstract
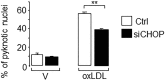
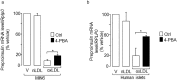
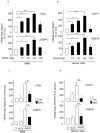
Elevated plasma concentration of the pro-atherogenic oxidized low density lipoprotein cholesterol (LDL) triggers adverse effects in pancreatic beta-cells and is associated with type 2 diabetes. Here, we investigated whether the endoplasmic reticulum (ER) stress is a key player coupling oxidative stress to beta-cell dysfunction and death elicited by human oxidized LDL. We found that human oxidized LDL activates ER stress as evidenced by the activation of the inositol requiring 1α, and the elevated expression of both DDIT3 (also called CHOP) and DNAJC3 (also called P58IPK) ER stress markers in isolated human islets and the mouse insulin secreting MIN6 cells. Silencing of Chop and inhibition of ER stress markers by the chemical chaperone phenyl butyric acid (PBA) prevented cell death caused by oxidized LDL. Finally, we found that oxidative stress accounts for activation of ER stress markers induced by oxidized LDL. Induction of Chop/CHOP and p58IPK/P58IPK by oxidized LDL was mimicked by hydrogen peroxide and was blocked by co-treatment with the N-acetylcystein antioxidant. As a conclusion, the harmful effects of oxidized LDL in beta-cells requires ER stress activation in a manner that involves oxidative stress. This mechanism may account for impaired beta-cell function in diabetes and can be reversed by antioxidant treatment.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




References
-
- Butler AE, Janson J, Bonner-Weir S, Ritzel R, Rizza RA, Butler PC. Beta-cell deficit and increased beta-cell apoptosis in humans with type 2 diabetes. Diabetes. 2003;52: 102–10. - PubMed
-
- Weir GC, Laybutt DR, Kaneto H, Bonner-Weir S, Sharma A. Beta-cell adaptation and decompensation during the progression of diabetes. Diabetes. 2001;50 Suppl 1: S154–9. - PubMed
-
- Rohrer L, Hersberger M, von Eckardstein A. High density lipoproteins in the intersection of diabetes mellitus, inflammation and cardiovascular disease. Curr Opin Lipidol. 2004;15: 269–78. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

