Chimeric Antigen Receptor T Cells and Hematopoietic Cell Transplantation: How Not to Put the CART Before the Horse
- PMID: 27638367
- PMCID: PMC5237606
- DOI: 10.1016/j.bbmt.2016.09.002
Chimeric Antigen Receptor T Cells and Hematopoietic Cell Transplantation: How Not to Put the CART Before the Horse
Abstract
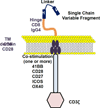
Hematopoietic cell transplantation (HCT) remains an important and potentially curative option for most hematologic malignancies. As a form of immunotherapy, allogeneic HCT (allo-HCT) offers the potential for durable remissions but is limited by transplantation- related morbidity and mortality owing to organ toxicity, infection, and graft-versus-host disease. The recent positive outcomes of chimeric antigen receptor T (CART) cell therapy in B cell malignancies may herald a paradigm shift in the management of these disorders and perhaps other hematologic malignancies as well. Clinical trials are now needed to address the relative roles of CART cells and HCT in the context of transplantation-eligible patients. In this review, we summarize the state of the art of the development of CART cell therapy for leukemia, lymphoma, and myeloma and discuss our perspective of how CART cell therapy can be applied in the context of HCT.
Keywords: Adoptive T cell therapy; Adoptive immunotherapy; Bone marrow transplantation; CART19; Chimeric antigen receptor T cells; Hematopoietic stem cell transplantation; Immunotherapy.
Copyright © 2017 The American Society for Blood and Marrow Transplantation. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors work under a research collaboration involving the University of Pennsylvania and the Novartis Institutes of Biomedical Research, Inc. S.S.K, D.L.P and S.G. are inventors of intellectual property licensed by the University of Pennsylvania to Novartis.
Figures


References
-
- Gill S, June CH. Going viral: chimeric antigen receptor T-cell therapy for hematological malignancies. Immunol Rev. 2015;263(1):68–89. - PubMed
-
- Milone MC, Fish JD, Carpenito C, Carroll RG, Binder GK, Teachey D, et al. Chimeric receptors containing CD137 signal transduction domains mediate enhanced survival of T cells and increased antileukemic efficacy in vivo. Molecular therapy : the journal of the American Society of Gene Therapy. 2009;17(8):1453–1464. - PMC - PubMed
-
- Pule MA, Straathof KC, Dotti G, Heslop HE, Rooney CM, Brenner MK. A chimeric T cell antigen receptor that augments cytokine release and supports clonal expansion of primary human T cells. Mol Ther. 2005;12(5):933–941. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

