Structural basis of synaptic vesicle assembly promoted by α-synuclein
- PMID: 27640673
- PMCID: PMC5031799
- DOI: 10.1038/ncomms12563
Structural basis of synaptic vesicle assembly promoted by α-synuclein
Erratum in
-
Corrigendum: Structural basis of synaptic vesicle assembly promoted by α-synuclein.Nat Commun. 2017 May 11;8:15667. doi: 10.1038/ncomms15667. Nat Commun. 2017. PMID: 28492274 Free PMC article. No abstract available.
Abstract
α-synuclein (αS) is an intrinsically disordered protein whose fibrillar aggregates are the major constituents of Lewy bodies in Parkinson's disease. Although the specific function of αS is still unclear, a general consensus is forming that it has a key role in regulating the process of neurotransmitter release, which is associated with the mediation of synaptic vesicle interactions and assembly. Here we report the analysis of wild-type αS and two mutational variants linked to familial Parkinson's disease to describe the structural basis of a molecular mechanism enabling αS to induce the clustering of synaptic vesicles. We provide support for this 'double-anchor' mechanism by rationally designing and experimentally testing a further mutational variant of αS engineered to promote stronger interactions between synaptic vesicles. Our results characterize the nature of the active conformations of αS that mediate the clustering of synaptic vesicles, and indicate their relevance in both functional and pathological contexts.
Conflict of interest statement
The authors declare no competing financial interests.
Figures
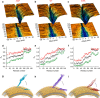

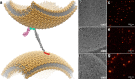

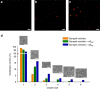
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

