Mechanosensory neurons control sweet sensing in Drosophila
- PMID: 27641708
- PMCID: PMC5031804
- DOI: 10.1038/ncomms12872
Mechanosensory neurons control sweet sensing in Drosophila
Abstract
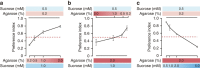
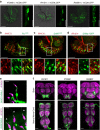
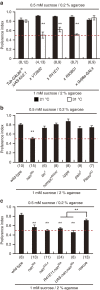
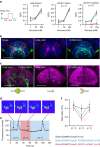
Animals discriminate nutritious food from toxic substances using their sense of taste. Since taste perception requires taste receptor cells to come into contact with water-soluble chemicals, it is a form of contact chemosensation. Concurrent with that contact, mechanosensitive cells detect the texture of food and also contribute to the regulation of feeding. Little is known, however, about the extent to which chemosensitive and mechanosensitive circuits interact. Here, we show Drosophila prefers soft food at the expense of sweetness and that this preference requires labellar mechanosensory neurons (MNs) and the mechanosensory channel Nanchung. Activation of these labellar MNs causes GABAergic inhibition of sweet-sensing gustatory receptor neurons, reducing the perceived intensity of a sweet stimulus. These findings expand our understanding of the ways different sensory modalities cooperate to shape animal behaviour.
Figures
Similar articles
-
A mechanosensory receptor required for food texture detection in Drosophila.Nat Commun. 2017 Jan 27;8:14192. doi: 10.1038/ncomms14192. Nat Commun. 2017. PMID: 28128210 Free PMC article.
-
Mechanosensory circuits coordinate two opposing motor actions in Drosophila feeding.Sci Adv. 2019 May 22;5(5):eaaw5141. doi: 10.1126/sciadv.aaw5141. eCollection 2019 May. Sci Adv. 2019. PMID: 31131327 Free PMC article.
-
The Basis of Food Texture Sensation in Drosophila.Neuron. 2016 Aug 17;91(4):863-877. doi: 10.1016/j.neuron.2016.07.013. Epub 2016 Jul 28. Neuron. 2016. PMID: 27478019 Free PMC article.
-
Taste perception, associated hormonal modulation, and nutrient intake.Nutr Rev. 2015 Feb;73(2):83-91. doi: 10.1093/nutrit/nuu009. Nutr Rev. 2015. PMID: 26024495 Free PMC article. Review.
-
Why do we like sweet taste: A bitter tale?Physiol Behav. 2016 Oct 1;164(Pt B):432-437. doi: 10.1016/j.physbeh.2016.05.007. Epub 2016 May 9. Physiol Behav. 2016. PMID: 27174610 Free PMC article. Review.
Cited by
-
Drosophila melanogaster Chemosensory Pathways as Potential Targets to Curb the Insect Menace.Insects. 2022 Jan 28;13(2):142. doi: 10.3390/insects13020142. Insects. 2022. PMID: 35206716 Free PMC article. Review.
-
Drosophila suzukii avoidance of microbes in oviposition choice.R Soc Open Sci. 2021 Jan 20;8(1):201601. doi: 10.1098/rsos.201601. eCollection 2021 Jan. R Soc Open Sci. 2021. PMID: 33614092 Free PMC article.
-
Diverse mechanisms of taste coding in Drosophila.Sci Adv. 2023 Nov 15;9(46):eadj7032. doi: 10.1126/sciadv.adj7032. Epub 2023 Nov 17. Sci Adv. 2023. PMID: 37976361 Free PMC article.
-
Drosophila Larval Light-Avoidance is Negatively Regulated by Temperature Through Two Pairs of Central Brain Neurons.Neurosci Bull. 2022 Feb;38(2):200-204. doi: 10.1007/s12264-021-00785-7. Epub 2021 Nov 9. Neurosci Bull. 2022. PMID: 34751918 Free PMC article. No abstract available.
-
Somatotopic organization among parallel sensory pathways that promote a grooming sequence in Drosophila.Elife. 2024 Apr 18;12:RP87602. doi: 10.7554/eLife.87602. Elife. 2024. PMID: 38634460 Free PMC article.
References
-
- Spence C. Multisensory flavor perception. Cell 161, 24–35 (2015). - PubMed
-
- Todrank J. & Bartoshuk L. M. A taste illusion: taste sensation localized by touch. Physiol. Behav. 50, 1027–1031 (1991). - PubMed
-
- Hollowood T. A., Linforth R. S. & Taylor A. J. The effect of viscosity on the perception of flavour. Chem. Senses 27, 583–591 (2002). - PubMed
-
- Tournier C. et al.. A study on texture-taste-aroma interactions: physico-chemical and cognitive mechanisms. Int. Dairy J. 19, 450–458 (2009).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

