Human CB1 Receptor Isoforms, present in Hepatocytes and β-cells, are Involved in Regulating Metabolism
- PMID: 27641999
- PMCID: PMC5027555
- DOI: 10.1038/srep33302
Human CB1 Receptor Isoforms, present in Hepatocytes and β-cells, are Involved in Regulating Metabolism
Abstract
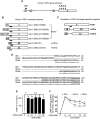
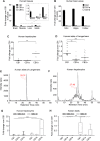
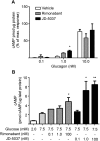
Therapeutics aimed at blocking the cannabinoid 1 (CB1) receptor for treatment of obesity resulted in significant improvements in liver function, glucose uptake and pancreatic β-cell function independent of weight loss or CB1 receptor blockade in the brain, suggesting that peripherally-acting only CB1 receptor blockers may be useful therapeutic agents. Neuropsychiatric side effects and lack of tissue specificity precluded clinical use of first-generation, centrally acting CB1 receptor blockers. In this study we specifically analyzed the potential relevance to diabetes of human CB1 receptor isoforms in extraneural tissues involved in glucose metabolism. We identified an isoform of the human CB1 receptor (CB1b) that is highly expressed in β-cells and hepatocytes but not in the brain. Importantly, CB1b shows stronger affinity for the inverse agonist JD-5037 than for rimonabant compared to CB1 full length. Most relevant to the field, CB1b is a potent regulator of adenylyl cyclase activity in peripheral metabolic tissues. CB1b blockade by JD-5037 results in stronger adenylyl cyclase activation compared to rimonabant and it is a better enhancer of insulin secretion in β-cells. We propose this isoform as a principal pharmacological target for the treatment of metabolic disorders involving glucose metabolism.
Figures


References
-
- Silvestri C. & Di Marzo V. The endocannabinoid system in energy homeostasis and the etiopathology of metabolic disorders. Cell Metab 17, 475–490 (2013). - PubMed
-
- Matias I. et al.. Regulation, function, and dysregulation of endocannabinoids in models of adipose and beta-pancreatic cells and in obesity and hyperglycemia. J. Clin. Endocrinol. Metab. 91, 3171–3180 (2006). - PubMed
-
- Sarzani R. et al.. Altered pattern of cannabinoid type 1 receptor expression in adipose tissue of dysmetabolic and overweight patients. Metabolism . 58, 361–367 (2009). - PubMed
-
- Ravinet Trillou C. et al.. Anti-obesity effect of SR141716, a CB1 receptor antagonist, in diet-induced obese mice. Am J Physiol Regul Integr Comp Physiol 284, R345–R353 (2003). - PubMed
-
- Despres J. P., Golay A. & Sjostrom L. Effects of rimonabant on metabolic risk factors in overweight patients with dyslipidemia. N Engl J Med 353, 2121–2134 (2005). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

