Full-length model of the human galectin-4 and insights into dynamics of inter-domain communication
- PMID: 27642006
- PMCID: PMC5027518
- DOI: 10.1038/srep33633
Full-length model of the human galectin-4 and insights into dynamics of inter-domain communication
Abstract
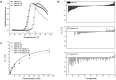
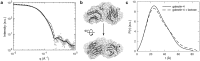
Galectins are proteins involved in diverse cellular contexts due to their capacity to decipher and respond to the information encoded by β-galactoside sugars. In particular, human galectin-4, normally expressed in the healthy gastrointestinal tract, displays differential expression in cancerous tissues and is considered a potential drug target for liver and lung cancer. Galectin-4 is a tandem-repeat galectin characterized by two carbohydrate recognition domains connected by a linker-peptide. Despite their relevance to cell function and pathogenesis, structural characterization of full-length tandem-repeat galectins has remained elusive. Here, we investigate galectin-4 using X-ray crystallography, small- and wide-angle X-ray scattering, molecular modelling, molecular dynamics simulations, and differential scanning fluorimetry assays and describe for the first time a structural model for human galectin-4. Our results provide insight into the structural role of the linker-peptide and shed light on the dynamic characteristics of the mechanism of carbohydrate recognition among tandem-repeat galectins.
Figures



Similar articles
-
Structural characterization of human galectin-4 C-terminal domain: elucidating the molecular basis for recognition of glycosphingolipids, sulfated saccharides and blood group antigens.FEBS J. 2015 Sep;282(17):3348-67. doi: 10.1111/febs.13348. Epub 2015 Jul 14. FEBS J. 2015. PMID: 26077389
-
An integrated computational analysis of the structure, dynamics, and ligand binding interactions of the human galectin network.J Chem Inf Model. 2011 Aug 22;51(8):1918-30. doi: 10.1021/ci200180h. Epub 2011 Jul 15. J Chem Inf Model. 2011. PMID: 21702482
-
Structure of the mouse galectin-4 N-terminal carbohydrate-recognition domain reveals the mechanism of oligosaccharide recognition.Acta Crystallogr D Biol Crystallogr. 2011 Mar;67(Pt 3):204-11. doi: 10.1107/S0907444911004082. Epub 2011 Feb 15. Acta Crystallogr D Biol Crystallogr. 2011. PMID: 21358051
-
Full-length galectin-8 and separate carbohydrate recognition domains: the whole is greater than the sum of its parts?Biochem Soc Trans. 2020 Jun 30;48(3):1255-1268. doi: 10.1042/BST20200311. Biochem Soc Trans. 2020. PMID: 32597487 Review.
-
Galectin-4 in normal tissues and cancer.Glycoconj J. 2004;20(4):247-55. doi: 10.1023/B:GLYC.0000025819.54723.a0. Glycoconj J. 2004. PMID: 15115909 Review.
Cited by
-
Insights into the recognition of hypermucoviscous Klebsiella pneumoniae clinical isolates by innate immune lectins of the Siglec and galectin families.Front Immunol. 2024 Aug 1;15:1436039. doi: 10.3389/fimmu.2024.1436039. eCollection 2024. Front Immunol. 2024. PMID: 39148735 Free PMC article.
-
Innate immune Galectin-7 specifically targets microbes that decorate themselves in blood group-like antigens.iScience. 2022 May 30;25(7):104482. doi: 10.1016/j.isci.2022.104482. eCollection 2022 Jul 15. iScience. 2022. PMID: 35754739 Free PMC article.
-
Semisynthesis and Biological Evaluation of Platencin Thioether Derivatives: Dual FabF and FabH Inhibitors against MRSA.ACS Med Chem Lett. 2021 Feb 15;12(3):433-442. doi: 10.1021/acsmedchemlett.0c00653. eCollection 2021 Mar 11. ACS Med Chem Lett. 2021. PMID: 33738071 Free PMC article.
-
Exploring functional pairing between surface glycoconjugates and human galectins using programmable glycodendrimersomes.Proc Natl Acad Sci U S A. 2018 Mar 13;115(11):E2509-E2518. doi: 10.1073/pnas.1720055115. Epub 2018 Jan 30. Proc Natl Acad Sci U S A. 2018. PMID: 29382751 Free PMC article.
-
Oligosaccharide Ligands of Galectin-4 and Its Subunits: Multivalency Scores Highly.Molecules. 2023 May 11;28(10):4039. doi: 10.3390/molecules28104039. Molecules. 2023. PMID: 37241779 Free PMC article.
References
-
- Barondes S. H., Cooper D. N., Gitt M. A. & Leffler H. Galectins. Structure and function of a large family of animal lectins. J Biol Chem 269, 20807–20810 (1994). - PubMed
-
- Hughes R. C. Secretion of the galectin family of mammalian carbohydrate-binding proteins. Biochim Biophys Acta 1473, 172–185 (1999). - PubMed
-
- Leffler H., Carlsson S., Hedlund M., Qian Y. & Poirier F. Introduction to galectins. Glycoconj J 19, 433–440 (2004). - PubMed
-
- Compagno D. et al.. Galectins: major signaling modulators inside and outside the cell. Curr Mol Med 14, 630–651 (2014). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

