PDE11A negatively regulates lithium responsivity
- PMID: 27646265
- PMCID: PMC5359083
- DOI: 10.1038/mp.2016.155
PDE11A negatively regulates lithium responsivity
Abstract
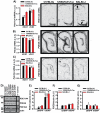
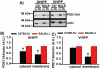
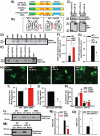
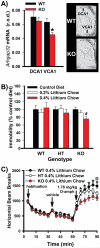
Lithium responsivity in patients with bipolar disorder has been genetically associated with Phosphodiesterase 11A (PDE11A), and lithium decreases PDE11A mRNA in induced pluripotent stem cell-derived hippocampal neurons originating from lithium-responsive patients. PDE11 is an enzyme uniquely enriched in the hippocampus that breaks down cyclic AMP and cyclic GMP. Here we determined whether decreasing PDE11A expression is sufficient to increase lithium responsivity in mice. In dorsal hippocampus and ventral hippocampus (VHIPP), lithium-responsive C57BL/6J and 129S6/SvEvTac mice show decreased PDE11A4 protein expression relative to lithium-unresponsive BALB/cJ mice. In VHIPP, C57BL/6J mice also show differences in PDE11A4 compartmentalization relative to BALB/cJ mice. In contrast, neither PDE2A nor PDE10A expression differ among the strains. The compartment-specific differences in PDE11A4 protein expression are explained by a coding single-nucleotide polymorphism (SNP) at amino acid 499, which falls within the GAF-B homodimerization domain. Relative to the BALB/cJ 499T, the C57BL/6J 499A decreases PDE11A4 homodimerization, which removes PDE11A4 from the membrane. Consistent with the observation that lower PDE11A4 expression correlates with better lithium responsiveness, we found that Pde11a knockout mice (KO) given 0.4% lithium chow for 3+ weeks exhibit greater lithium responsivity relative to wild-type (WT) littermates in tail suspension, an antidepressant-predictive assay, and amphetamine hyperlocomotion, an anti-manic predictive assay. Reduced PDE11A4 expression may represent a lithium-sensitive pathophysiology, because both C57BL/6J and Pde11a KO mice show increased expression of the pro-inflammatory cytokine interleukin-6 (IL-6) relative to BALB/cJ and PDE11A WT mice, respectively. Our finding that PDE11A4 negatively regulates lithium responsivity in mice suggests that the PDE11A SNPs identified in patients may be functionally relevant.
Figures

Similar articles
-
Conserved age-related increases in hippocampal PDE11A4 cause unexpected proteinopathies and cognitive decline of social associative memories.Aging Cell. 2022 Oct;21(10):e13687. doi: 10.1111/acel.13687. Epub 2022 Sep 8. Aging Cell. 2022. PMID: 36073342 Free PMC article.
-
PDE11A regulates social behaviors and is a key mechanism by which social experience sculpts the brain.Neuroscience. 2016 Oct 29;335:151-69. doi: 10.1016/j.neuroscience.2016.08.019. Epub 2016 Aug 17. Neuroscience. 2016. PMID: 27544407 Free PMC article.
-
A Role for Phosphodiesterase 11A (PDE11A) in the Formation of Social Memories and the Stabilization of Mood.Adv Neurobiol. 2017;17:201-230. doi: 10.1007/978-3-319-58811-7_8. Adv Neurobiol. 2017. PMID: 28956334 Free PMC article. Review.
-
Phosphodiesterase 11A (PDE11A), Enriched in Ventral Hippocampus Neurons, is Required for Consolidation of Social but not Nonsocial Memories in Mice.Neuropsychopharmacology. 2016 Nov;41(12):2920-2931. doi: 10.1038/npp.2016.106. Epub 2016 Jun 24. Neuropsychopharmacology. 2016. PMID: 27339393 Free PMC article.
-
Does phosphodiesterase 11A (PDE11A) hold promise as a future therapeutic target?Curr Pharm Des. 2015;21(3):389-416. doi: 10.2174/1381612820666140826114941. Curr Pharm Des. 2015. PMID: 25159071 Review.
Cited by
-
Conserved age-related increases in hippocampal PDE11A4 cause unexpected proteinopathies and cognitive decline of social associative memories.Aging Cell. 2022 Oct;21(10):e13687. doi: 10.1111/acel.13687. Epub 2022 Sep 8. Aging Cell. 2022. PMID: 36073342 Free PMC article.
-
Age-Related Increases in PDE11A4 Protein Expression Trigger Liquid-Liquid Phase Separation (LLPS) of the Enzyme That Can Be Reversed by PDE11A4 Small Molecule Inhibitors.Cells. 2025 Jun 13;14(12):897. doi: 10.3390/cells14120897. Cells. 2025. PMID: 40558524 Free PMC article.
-
PDE11A regulates social behaviors and is a key mechanism by which social experience sculpts the brain.Neuroscience. 2016 Oct 29;335:151-69. doi: 10.1016/j.neuroscience.2016.08.019. Epub 2016 Aug 17. Neuroscience. 2016. PMID: 27544407 Free PMC article.
-
A genetic basis for friendship? Homophily for membrane-associated PDE11A-cAMP-CREB signaling in CA1 of hippocampus dictates mutual social preference in male and female mice.Mol Psychiatry. 2021 Dec;26(12):7107-7117. doi: 10.1038/s41380-021-01237-4. Epub 2021 Jul 28. Mol Psychiatry. 2021. PMID: 34321593 Free PMC article.
-
Therapeutic targeting of 3',5'-cyclic nucleotide phosphodiesterases: inhibition and beyond.Nat Rev Drug Discov. 2019 Oct;18(10):770-796. doi: 10.1038/s41573-019-0033-4. Epub 2019 Aug 6. Nat Rev Drug Discov. 2019. PMID: 31388135 Free PMC article. Review.
References
-
- Malhi GS, Tanious M, Das P, Berk M. The science and practice of lithium therapy. Aust N Z J Psychiatry. 2012;46:192–211. - PubMed
-
- Yuasa K, Ohgaru T, Asahina M, Omori K. Identification of rat cyclic nucleotide phosphodiesterase 11A (PDE11A): comparison of rat and human PDE11A splicing variants. Eur J Biochem. 2001;268:4440–8. - PubMed
-
- Yuasa K, Kanoh Y, Okumura K, Omori K. Genomic organization of the human phosphodiesterase PDE11A gene. Evolutionary relatedness with other PDEs containing GAF domains. Eur J Biochem. 2001;268:168–78. - PubMed
-
- Kelly MP, Adamowicz W, Bove S, Hartman AJ, Mariga A, Pathak G, et al. Select 3',5'-cyclic nucleotide phosphodiesterases exhibit altered expression in the aged rodent brain. Cell Signal. 2014;26:383–97. - PubMed
-
- Kelly MP. Does phosphodiesterase 11A (PDE11A) hold promise as a future therapeutic target? Curr Pharm Des. 2015;21:389–416. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

