Ultradonut topology of the nuclear envelope
- PMID: 27647910
- PMCID: PMC5056109
- DOI: 10.1073/pnas.1604777113
Ultradonut topology of the nuclear envelope
Abstract
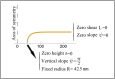
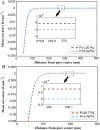
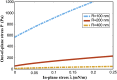
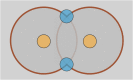
The nuclear envelope is a unique topological structure formed by lipid membranes in eukaryotic cells. Unlike other membrane structures, the nuclear envelope comprises two concentric membrane shells fused at numerous sites with toroid-shaped pores that impart a "geometric" genus on the order of thousands. Despite the intriguing architecture and vital biological functions of the nuclear membranes, how they achieve and maintain such a unique arrangement remains unknown. Here, we used the theory of elasticity and differential geometry to analyze the equilibrium shape and stability of this structure. Our results show that modest in- and out-of-plane stresses present in the membranes not only can define the pore geometry, but also provide a mechanism for destabilizing membranes beyond a critical size and set the stage for the formation of new pores. Our results suggest a mechanism wherein nanoscale buckling instabilities can define the global topology of a nuclear envelope-like structure.
Keywords: buckling instability; lipid membranes; nuclear envelope; topology.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







Similar articles
-
Isolation and fractionation of rat liver nuclear envelopes and nuclear pore complexes.Methods. 2006 Aug;39(4):277-83. doi: 10.1016/j.ymeth.2006.06.003. Methods. 2006. PMID: 16870471 Review.
-
Breakage of the nuclear envelope by an extending mitotic nucleus occurs during anaphase in Schizosaccharomyces japonicus.Genes Cells. 2011 Sep;16(9):911-26. doi: 10.1111/j.1365-2443.2011.01540.x. Epub 2011 Jul 6. Genes Cells. 2011. PMID: 21733045
-
Structural analysis of the nuclear pore complex by integrated approaches.Curr Opin Struct Biol. 2009 Apr;19(2):226-32. doi: 10.1016/j.sbi.2009.02.009. Epub 2009 Mar 25. Curr Opin Struct Biol. 2009. PMID: 19327984 Review.
-
3D ultrastructure of the nuclear pore complex.Curr Opin Cell Biol. 2012 Feb;24(1):86-91. doi: 10.1016/j.ceb.2011.12.011. Epub 2012 Jan 11. Curr Opin Cell Biol. 2012. PMID: 22244612 Free PMC article. Review.
-
Modularity within the architecture of the nuclear pore complex.Curr Opin Struct Biol. 2005 Apr;15(2):221-6. doi: 10.1016/j.sbi.2005.03.003. Curr Opin Struct Biol. 2005. PMID: 15837182 Review.
Cited by
-
Melatonin: Regulation of Biomolecular Condensates in Neurodegenerative Disorders.Antioxidants (Basel). 2021 Sep 17;10(9):1483. doi: 10.3390/antiox10091483. Antioxidants (Basel). 2021. PMID: 34573116 Free PMC article. Review.
-
Components and Mechanisms of Nuclear Mechanotransduction.Annu Rev Cell Dev Biol. 2021 Oct 6;37:233-256. doi: 10.1146/annurev-cellbio-120319-030049. Epub 2021 Jul 2. Annu Rev Cell Dev Biol. 2021. PMID: 34213953 Free PMC article. Review.
-
A Review of Continuum Mechanics for Mechanical Deformation of Lipid Membranes.Membranes (Basel). 2023 May 3;13(5):493. doi: 10.3390/membranes13050493. Membranes (Basel). 2023. PMID: 37233554 Free PMC article. Review.
-
Modelling Nuclear Morphology and Shape Transformation: A Review.Membranes (Basel). 2021 Jul 16;11(7):540. doi: 10.3390/membranes11070540. Membranes (Basel). 2021. PMID: 34357190 Free PMC article. Review.
-
Recapitulation of molecular regulators of nuclear motion during cell migration.Cell Adh Migr. 2019 Dec;13(1):50-62. doi: 10.1080/19336918.2018.1506654. Epub 2018 Sep 27. Cell Adh Migr. 2019. PMID: 30261154 Free PMC article. Review.
References
-
- D’Angelo MA, Anderson DJ, Richard E, Hetzer MW. Nuclear pores form de novo from both sides of the nuclear envelope. Science. 2006;312(5772):440–443. - PubMed
-
- Kreyszig E. Differential Geometry. Dover Publications, Inc.; Mineola, NY: 1991. pp. 172–174.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

