Biophysical comparison of ATP synthesis mechanisms shows a kinetic advantage for the rotary process
- PMID: 27647911
- PMCID: PMC5056049
- DOI: 10.1073/pnas.1608533113
Biophysical comparison of ATP synthesis mechanisms shows a kinetic advantage for the rotary process
Abstract
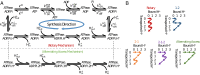
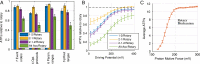
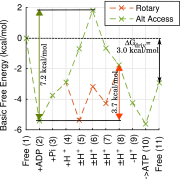
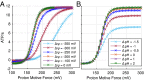
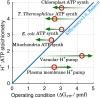
The ATP synthase (F-ATPase) is a highly complex rotary machine that synthesizes ATP, powered by a proton electrochemical gradient. Why did evolution select such an elaborate mechanism over arguably simpler alternating-access processes that can be reversed to perform ATP synthesis? We studied a systematic enumeration of alternative mechanisms, using numerical and theoretical means. When the alternative models are optimized subject to fundamental thermodynamic constraints, they fail to match the kinetic ability of the rotary mechanism over a wide range of conditions, particularly under low-energy conditions. We used a physically interpretable, closed-form solution for the steady-state rate for an arbitrary chemical cycle, which clarifies kinetic effects of complex free-energy landscapes. Our analysis also yields insights into the debated "kinetic equivalence" of ATP synthesis driven by transmembrane pH and potential difference. Overall, our study suggests that the complexity of the F-ATPase may have resulted from positive selection for its kinetic advantage.
Keywords: ATP synthase; evolution; free-energy landscape; kinetic mechanism; nonequilibrium steady state.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
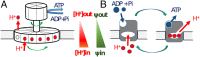
Similar articles
-
Biophysical comparison of ATP-driven proton pumping mechanisms suggests a kinetic advantage for the rotary process depending on coupling ratio.PLoS One. 2017 Mar 20;12(3):e0173500. doi: 10.1371/journal.pone.0173500. eCollection 2017. PLoS One. 2017. PMID: 28319179 Free PMC article.
-
Determination of the partial reactions of rotational catalysis in F1-ATPase.Biochemistry. 2007 Jul 31;46(30):8785-97. doi: 10.1021/bi700610m. Epub 2007 Jul 10. Biochemistry. 2007. PMID: 17620014
-
Torque-coupled thermodynamic model for F_{o}F_{1}-ATPase.Phys Rev E. 2017 May;95(5-1):052413. doi: 10.1103/PhysRevE.95.052413. Epub 2017 May 23. Phys Rev E. 2017. PMID: 28618520
-
Torque, chemistry and efficiency in molecular motors: a study of the rotary-chemical coupling in F1-ATPase.Q Rev Biophys. 2015 Nov;48(4):395-403. doi: 10.1017/S0033583515000050. Q Rev Biophys. 2015. PMID: 26537397 Free PMC article. Review.
-
The mechanism of rotating proton pumping ATPases.Biochim Biophys Acta. 2010 Aug;1797(8):1343-52. doi: 10.1016/j.bbabio.2010.02.014. Epub 2010 Feb 17. Biochim Biophys Acta. 2010. PMID: 20170625 Review.
Cited by
-
Biophysical comparison of ATP-driven proton pumping mechanisms suggests a kinetic advantage for the rotary process depending on coupling ratio.PLoS One. 2017 Mar 20;12(3):e0173500. doi: 10.1371/journal.pone.0173500. eCollection 2017. PLoS One. 2017. PMID: 28319179 Free PMC article.
-
Poor Person's pH Simulation of Membrane Proteins.Methods Mol Biol. 2021;2315:197-217. doi: 10.1007/978-1-0716-1468-6_12. Methods Mol Biol. 2021. PMID: 34302678
-
Opposing Pressures of Speed and Efficiency Guide the Evolution of Molecular Machines.Mol Biol Evol. 2019 Dec 1;36(12):2813-2822. doi: 10.1093/molbev/msz190. Mol Biol Evol. 2019. PMID: 31432071 Free PMC article.
-
Allocating dissipation across a molecular machine cycle to maximize flux.Proc Natl Acad Sci U S A. 2017 Oct 17;114(42):11057-11062. doi: 10.1073/pnas.1707534114. Epub 2017 Oct 3. Proc Natl Acad Sci U S A. 2017. PMID: 29073016 Free PMC article.
-
Multi-scale reactor designs extend the physical limits of fixation.bioRxiv [Preprint]. 2024 Aug 30:2024.08.28.610213. doi: 10.1101/2024.08.28.610213. bioRxiv. 2024. PMID: 39257791 Free PMC article. Preprint.
References
-
- Mitchell P. Coupling of phosphorylation to electron and hydrogen transfer by a chemi-osmotic type of mechanism. Nature. 1961;191:144–148. - PubMed
-
- Boyer PD. The binding change mechanism for ATP synthase—some probabilities and possibilities. Biochim Biophys Acta. 1993;1140(3):215–250. - PubMed
-
- Cross RL, Müller V. The evolution of A-, F-, and V-type ATP synthases and ATPases: Reversals in function and changes in the H+/ATP coupling ratio. FEBS Lett. 2004;576(1-2):1–4. - PubMed
-
- Grüber G, Manimekalai MS, Mayer F, Müller V. ATP synthases from archaea: The beauty of a molecular motor. Biochim Biophys Acta Bioenerg. 2014;1837(6):940–952. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

