Cellulose synthase complexes act in a concerted fashion to synthesize highly aggregated cellulose in secondary cell walls of plants
- PMID: 27647923
- PMCID: PMC5056089
- DOI: 10.1073/pnas.1613273113
Cellulose synthase complexes act in a concerted fashion to synthesize highly aggregated cellulose in secondary cell walls of plants
Abstract
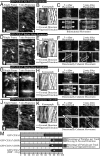
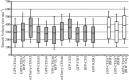
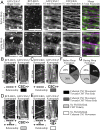
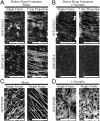
Cellulose, often touted as the most abundant biopolymer on Earth, is a critical component of the plant cell wall and is synthesized by plasma membrane-spanning cellulose synthase (CESA) enzymes, which in plants are organized into rosette-like CESA complexes (CSCs). Plants construct two types of cell walls, primary cell walls (PCWs) and secondary cell walls (SCWs), which differ in composition, structure, and purpose. Cellulose in PCWs and SCWs is chemically identical but has different physical characteristics. During PCW synthesis, multiple dispersed CSCs move along a shared linear track in opposing directions while synthesizing cellulose microfibrils with low aggregation. In contrast, during SCW synthesis, we observed swaths of densely arranged CSCs that moved in the same direction along tracks while synthesizing cellulose microfibrils that became highly aggregated. Our data support a model in which distinct spatiotemporal features of active CSCs during PCW and SCW synthesis contribute to the formation of cellulose with distinct structure and organization in PCWs and SCWs of Arabidopsis thaliana This study provides a foundation for understanding differences in the formation, structure, and organization of cellulose in PCWs and SCWs.
Keywords: cellulose microfibrils; cellulose synthase complex; live-cell imaging; plasma membrane; protein dynamics.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Identifying Stable Fragments of Arabidopsis thaliana Cellulose Synthase Subunit 3 by Yeast Display.Biotechnol J. 2019 Apr;14(4):e1800353. doi: 10.1002/biot.201800353. Epub 2018 Sep 25. Biotechnol J. 2019. PMID: 30171735
-
Cellulose synthesis in higher plants.Annu Rev Cell Dev Biol. 2006;22:53-78. doi: 10.1146/annurev.cellbio.22.022206.160206. Annu Rev Cell Dev Biol. 2006. PMID: 16824006 Review.
-
Organization of cellulose synthase complexes involved in primary cell wall synthesis in Arabidopsis thaliana.Proc Natl Acad Sci U S A. 2007 Sep 25;104(39):15572-7. doi: 10.1073/pnas.0706569104. Epub 2007 Sep 18. Proc Natl Acad Sci U S A. 2007. PMID: 17878303 Free PMC article.
-
Formation of wood secondary cell wall may involve two type cellulose synthase complexes in Populus.Plant Mol Biol. 2017 Mar;93(4-5):419-429. doi: 10.1007/s11103-016-0570-8. Epub 2016 Dec 16. Plant Mol Biol. 2017. PMID: 27987127
-
Cellulose biosynthesis in plants: from genes to rosettes.Plant Cell Physiol. 2002 Dec;43(12):1407-20. doi: 10.1093/pcp/pcf164. Plant Cell Physiol. 2002. PMID: 12514238 Review.
Cited by
-
Tissue-specific directionality of cellulose synthase complex movement inferred from cellulose microfibril polarity in secondary cell walls of Arabidopsis.Sci Rep. 2023 Dec 12;13(1):22007. doi: 10.1038/s41598-023-48545-z. Sci Rep. 2023. PMID: 38086837 Free PMC article.
-
Two Complementary Mechanisms Underpin Cell Wall Patterning during Xylem Vessel Development.Plant Cell. 2017 Oct;29(10):2433-2449. doi: 10.1105/tpc.17.00309. Epub 2017 Sep 25. Plant Cell. 2017. PMID: 28947492 Free PMC article.
-
A Structurally Specialized Uniform Wall Layer is Essential for Constructing Wall Ingrowth Papillae in Transfer Cells.Front Plant Sci. 2017 Dec 5;8:2035. doi: 10.3389/fpls.2017.02035. eCollection 2017. Front Plant Sci. 2017. PMID: 29259611 Free PMC article.
-
Exploiting CELLULOSE SYNTHASE (CESA) Class Specificity to Probe Cellulose Microfibril Biosynthesis.Plant Physiol. 2018 May;177(1):151-167. doi: 10.1104/pp.18.00263. Epub 2018 Mar 9. Plant Physiol. 2018. PMID: 29523715 Free PMC article.
-
Lignin-polysaccharide interactions in plant secondary cell walls revealed by solid-state NMR.Nat Commun. 2019 Jan 21;10(1):347. doi: 10.1038/s41467-018-08252-0. Nat Commun. 2019. PMID: 30664653 Free PMC article.
References
-
- Cosgrove DJ. Growth of the plant cell wall. Nat Rev Mol Cell Biol. 2005;6(11):850–861. - PubMed
-
- Kumar M, Campbell L, Turner S. Secondary cell walls: Biosynthesis and manipulation. J Exp Bot. 2016;67(2):515–531. - PubMed
-
- Pauly M, Keegstra K. Biosynthesis of the plant cell wall matrix polysaccharide xyloglucan. Annu Rev Plant Biol. 2016;67:235–259. - PubMed
-
- Anderson CT. We be jammin’: An update on pectin biosynthesis, trafficking and dynamics. J Exp Bot. 2016;67(2):495–502. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

