Independent Control of Elastomer Properties through Stereocontrolled Synthesis
- PMID: 27654023
- PMCID: PMC5082520
- DOI: 10.1002/anie.201606750
Independent Control of Elastomer Properties through Stereocontrolled Synthesis
Abstract
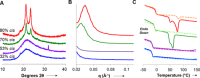
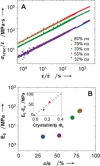
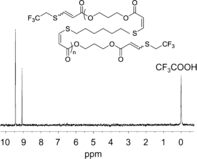
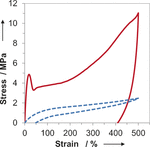
In most synthetic elastomers, changing the physical properties by monomer choice also results in a change to the crystallinity of the material, which manifests through alteration of its mechanical performance. Using organocatalyzed stereospecific additions of thiols to activated alkynes, high-molar-mass elastomers were isolated via step-growth polymerization. The resulting controllable double-bond stereochemistry defines the crystallinity and the concomitant mechanical properties as well as enabling the synthesis of materials that retain their excellent mechanical properties through changing monomer composition. Using this approach to elastomer synthesis, further end group modification and toughening through vulcanization strategies are also possible. The organocatalytic control of stereochemistry opens the realm to a new and easily scalable class of elastomers that will have unique chemical handles for functionalization and post synthetic processing.
Keywords: click chemistry; elastomers; organocatalysis; step-growth polymerization; stereochemistry.
© 2016 The Authors. Published by Wiley-VCH Verlag GmbH & Co. KGaA.
Figures


References
-
- None
-
- Holden G., Kricheldorf H. R., Quirk R. P., 3rd ed. Thermoplastic Elastomers, Hanser, Munich, 2004;
-
- Krol P., Prog. Mater. Sci. 2007, 52, 915–1015;
-
- Spontak R. J., Patel N. P., Curr. Opin. Colloid Interface Sci. 2000, 5, 334–341.
-
- None
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

