Single fixed-dose oral dexketoprofen plus tramadol for acute postoperative pain in adults
- PMID: 27654994
- PMCID: PMC6457609
- DOI: 10.1002/14651858.CD012232.pub2
Single fixed-dose oral dexketoprofen plus tramadol for acute postoperative pain in adults
Abstract
Background: Combining two different analgesics in fixed doses in a single tablet can provide better pain relief than either drug alone in acute pain. This appears to be broadly true across a range of different drug combinations, in postoperative pain and migraine headache. A new combination of dexketoprofen (a nonsteroidal anti-inflammatory drug) plus tramadol (an opioid) has been tested in acute postoperative pain conditions. It is not yet licensed for use. This review is one of a series on oral analgesics for acute postoperative pain. Individual reviews have been brought together in two overviews to provide information about the relative efficacy and harm of the different interventions.
Objectives: To assess the analgesic efficacy and adverse effects of a single fixed-dose of oral dexketoprofen plus tramadol, compared with placebo, for moderate to severe postoperative pain in adults, using methods that permit comparison with other analgesics evaluated in standardised trials using almost identical methods and outcomes. A secondary objective was to compare the combination with the individual analgesics alone.
Search methods: We searched the Cochrane Central Register of Controlled Trials (CENTRAL) via CRSO, MEDLINE via Ovid, and Embase via Ovid from inception to 31 May 2016. We also searched the reference lists of retrieved studies and reviews, and two online clinical trial registries.
Selection criteria: Randomised, double-blind trials of oral dexketoprofen plus tramadol administered as a single oral dose, for the relief of acute postoperative pain in adults, and compared to placebo.
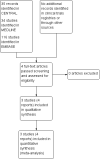
Data collection and analysis: Two review authors independently considered trials for inclusion in the review, examined issues of study quality and potential bias, and extracted data. For dichotomous outcomes, we calculated risk ratio (RR) and number needed to treat for an additional beneficial outcome (NNT) for dexketoprofen plus tramadol, compared with placebo with 95% confidence intervals (CI). We collected information on the number of participants with at least 50% of the maximum possible pain relief over six hours, the median time to use of rescue medication, and the proportion of participants requiring rescue medication. We also collected information on adverse events and withdrawals. We assessed the quality of the evidence using GRADE and created a 'Summary of findings' table.We also collected information on the number of participants with at least 50% of the maximum possible pain relief over six hours for dexketoprofen alone and tramadol alone.
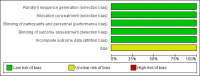
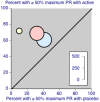
Main results: We included three studies with 1853 participants who had undergone surgical removal of impacted wisdom teeth, hip replacement, or hysterectomy. The overall risk of bias across the three included studies was low, with unclear risk of bias in relation to the size of the three studies. Two studies did not report all our prespecified outcomes, which limited the analyses we could do.The proportion of participants achieving at least 50% pain relief over six hours with dexketoprofen 25 mg plus tramadol 75 mg was 66%, compared to 32% with placebo, giving an NNT of 3.0 (95% CI 2.5 to 3.7) (RR 2.1 (95% CI 1.7 to 2.4); 748 participants; 3 studies) (moderate quality evidence). The response rate with dexketoprofen 25 mg alone was 53% (RR 1.3 (95% CI 1.1 to 1.4); 744 participants; 3 studies) and with tramadol alone was 45% (RR 1.5 (95% CI 1.3 to 1.7); 741 participants; 3 studies) (moderate quality evidence). We downgraded the evidence because of some inconsistency in the results.The median time to use of rescue medication could not be estimated exactly, but was probably eight hours or more, indicating a long duration of effect (moderate quality evidence). We downgraded the evidence because it was not possible to estimate the effect exactly in the two multiple dose studies, resulting in imprecision. Fewer participants used rescue medication with higher doses of active treatment (summary statistic not calculated; 123 participants; 1 study) (very low quality evidence). We downgraded the evidence because the data came from a single study with few participants and events.Adverse events and serious adverse events were not reported consistently for the single dose phase of the studies. In the single dose study, 11% of participants experienced adverse events with dexketoprofen 25 mg plus tramadol 75 mg, which were mostly mild or moderate nausea, vomiting, or dizziness, and typical with these medicines. Rates were lower with placebo and lower doses (very low quality evidence). We downgraded the evidence because the data came from a single study with few participants and events. Information on multiple dosing over three and five days supported a low event rate with the combination. Overall, rates were generally low in all treatment arms, as they were for withdrawals for adverse events or other reasons.
Authors' conclusions: A single oral dose of dexketoprofen 25 mg plus tramadol 75 mg provided good levels of pain relief with long duration of action to more people than placebo or the same dose of dexketoprofen or tramadol alone. The magnitude of the effect was similar to other good analgesics. Adverse event rates were low.There is modest uncertainty about the precision of the point estimate for efficacy, but the NNT of 3 is consistent with other analgesics considered effective and commonly used.
Conflict of interest statement
SD: none known.
TC: none known.
TP: none known; TP is an anaesthetist and manages patients with acute pain.
Figures

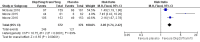



Update of
- doi: 10.1002/14651858.CD012232
Similar articles
-
Single-dose intravenous ketorolac for acute postoperative pain in adults.Cochrane Database Syst Rev. 2021 May 17;5(5):CD013263. doi: 10.1002/14651858.CD013263.pub2. Cochrane Database Syst Rev. 2021. PMID: 33998669 Free PMC article.
-
Tramadol for osteoarthritis.Cochrane Database Syst Rev. 2019 May 27;5(5):CD005522. doi: 10.1002/14651858.CD005522.pub3. Cochrane Database Syst Rev. 2019. PMID: 31132298 Free PMC article.
-
Pharmacological interventions for painful sickle cell vaso-occlusive crises in adults.Cochrane Database Syst Rev. 2019 Nov 14;2019(11):CD012187. doi: 10.1002/14651858.CD012187.pub2. Cochrane Database Syst Rev. 2019. PMID: 31742673 Free PMC article.
-
Perioperative intravenous ketamine for acute postoperative pain in adults.Cochrane Database Syst Rev. 2018 Dec 20;12(12):CD012033. doi: 10.1002/14651858.CD012033.pub4. Cochrane Database Syst Rev. 2018. PMID: 30570761 Free PMC article.
-
Ketoprofen for episodic tension-type headache in adults.Cochrane Database Syst Rev. 2016 Sep 22;9(9):CD012190. doi: 10.1002/14651858.CD012190.pub2. Cochrane Database Syst Rev. 2016. PMID: 27654948 Free PMC article. Review.
Cited by
-
Advances in the Management of Acute Postsurgical Pain: A Review.Cureus. 2023 Aug 4;15(8):e42974. doi: 10.7759/cureus.42974. eCollection 2023 Aug. Cureus. 2023. PMID: 37671225 Free PMC article. Review.
-
Expert Consensus on Clinical Use of an Orally Administered Dexketoprofen Plus Tramadol Fixed-Dose Combination in Moderate-To-Severe Acute Pain: A Delphi Study.Adv Ther. 2019 Nov;36(11):3174-3185. doi: 10.1007/s12325-019-01096-0. Epub 2019 Sep 18. Adv Ther. 2019. PMID: 31535328 Free PMC article.
-
Novel Pharmacological Nonopioid Therapies in Chronic Pain.Curr Pain Headache Rep. 2018 Apr 3;22(4):31. doi: 10.1007/s11916-018-0674-8. Curr Pain Headache Rep. 2018. PMID: 29616344 Review.
-
A "novel" association to treat pain: tramadol/dexketoprofen. The first drug of a "new pharmacological class".Acta Biomed. 2017 Apr 28;88(1):17-24. doi: 10.23750/abm.v88i1.6361. Acta Biomed. 2017. PMID: 28467329 Free PMC article.
-
Risk Factors and Prevention Strategies for Postoperative Opioid Abuse.Pain Res Manag. 2019 Jul 10;2019:7490801. doi: 10.1155/2019/7490801. eCollection 2019. Pain Res Manag. 2019. PMID: 31360271 Free PMC article. Review.
References
References to studies included in this review
McQuay 2016 {published and unpublished data}
-
- McQuay HJ, Moore RA, Berta A, Gainutdinovs O, Fulesdi B, Porvaneckas N, et al. Randomized clinical trial of dexketoprofen/tramadol 25 mg/75 mg in moderate‐to‐severe pain after total hip arthroplasty. Revista de la Sociedad Espanola del Dolor 2015;22(5):186‐99. - PubMed
-
- McQuay HJ, Moore RA, Berta A, Gainutdinovs O, Fülesdi B, Porvaneckas N, et al. Randomized clinical trial of dexketoprofen/tramadol 25 mg/75 mg in moderate‐to‐severe pain after total hip arthroplasty. British Journal of Anaesthesia 2016;116(2):269‐76. [CTG: NCT01902134; DOI: 10.1093/bja/aev457] - DOI - PMC - PubMed
Moore 2015 {published data only}
-
- Moore RA, Gay‐Escoda C, Figueiredo R, Tóth‐Bagi Z, Dietrich T, Milleri S, et al. Dexketoprofen/tramadol: randomised double‐blind trial and confirmation of empirical theory of combination analgesics in acute pain. Journal of Headache and Pain 2015;16:541. [CTG: NCT01307020; DOI: 10.1186/s10194-015-0541-5] - DOI - PMC - PubMed
Moore 2016 {published data only (unpublished sought but not used)}
-
- Moore RA, McQuay HJ, Tomaszewski J, Raba G, Tutunaru D, Lietuviete N, et al. Dexketoprofen/tramadol 25 mg/75 mg: randomised double‐blind trial in moderate‐to‐severe acute pain after abdominal hysterectomy. BMC Anesthesiology 2016;16:9. [CTG: NCT01904149; DOI: 10.1186/s12871-016-0174-5] - DOI - PMC - PubMed
Additional references
Barbanoj 2001
Barden 2004
Barden 2006
Barden 2009
Chaparro 2012
Collins 1997
Collins 2001
Cook 1995
Cooper 1991
-
- Cooper SA. Single‐dose analgesic studies: the upside and downside of assay sensitivity. In: Max MB, Portenoy RK, Laska EM editor(s). The Design of Analgesic Clinical Trials. Advances in Pain Research and Therapy. Vol. 18, New York: Raven Press, 1991:117‐24.
Derry 2009
Derry 2010
Derry 2013
Derry 2015
FitzGerald 2001
GRADEpro GDT 2015 [Computer program]
-
- McMaster University. GRADEpro Guideline Development Tool. McMaster University, 2015.
Grond 2004
Guyatt 2013a
-
- Guyatt G, Oxman AD, Sultan S, Brozek J, Glasziou P, Alonso‐Coello P, et al. GRADE guidelines: 11. Making an overall rating of confidence in effect estimates for a single outcome and for all outcomes. Journal of Clinical Epidemiology 2013;66(2):151‐7. [DOI: 10.1016/j.jclinepi.2012.01.006] - DOI - PubMed
Guyatt 2013b
Hernández‐Díaz 2000
-
- Hernández‐Díaz S, García Rodríguez LA. Association between nonsteroidal anti‐inflammatory drugs and upper gastrointestinal tract bleeding/perforation: an overview of epidemiologic studies published in the 1990s. Archives of Internal Medicine 2000;160:2093‐9. [DOI: 10.1001/archinte.160.14.2093] - DOI - PubMed
Herrero 2003
Higgins 2011
-
- Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org.
L'Abbé 1987
Laporte 2004
Lassen 2015
McQuay 2005
McQuay 2012
Menarini 2016
-
- Dexketoprofen+Tramadol. www.menarini.com/Home/Innovation‐Research/Our‐research/Dexketoprofen‐Tra... (accessed 3 June 2016).
Moher 2009
Moore 1996
Moore 1997a
Moore 1997b
Moore 1998
Moore 2003
-
- Moore RA, Edwards J, Barden J, McQuay HJ. Bandolier's Little Book of Pain. Oxford: Oxford University Press, 2003. [ISBN: 0‐19‐263247‐7]
Moore 2005
Moore 2006
-
- Moore A, McQuay H. Bandolier's Little Book of Making Sense of the Medical Evidence. Oxford: Oxford University Press, 2006. [ISBN: 0‐19‐856604‐2]
Moore 2008a
Moore 2008b
-
- Moore RA, Barden J, Derry S, McQuay HJ. Managing potential publication bias. In: McQuay HJ, Kalso E, Moore RA editor(s). Systematic Reviews in Pain Research: Methodology Refined. Seattle: IASP Press, 2008:15‐24. [ISBN: 978‐0‐931092‐69‐5]
Moore 2011
-
- Moore RA, Straube S, Paine J, Derry S, McQuay HJ. Minimum efficacy criteria for comparisons between treatments using individual patient meta‐analysis of acute pain trials: examples of etoricoxib, paracetamol, ibuprofen, and ibuprofen/paracetamol combinations after third molar extraction. Pain 2011;152(5):982‐9. [DOI: 10.1016/j.pain.2010.11.030] - DOI - PubMed
Moore 2012
Moore 2015a
Moore 2015b
PaPaS 2012
-
- Cochrane Pain, Palliative and Supportive Care Group. PaPaS author and referee guidance. papas.cochrane.org/papas‐documents (accessed 28 April 2016).
RevMan 2014 [Computer program]
-
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager. Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Toms 2008
Toms 2009
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

