Degradation Rate of 5-Fluorouracil in Metastatic Colorectal Cancer: A New Predictive Outcome Biomarker?
- PMID: 27656891
- PMCID: PMC5033390
- DOI: 10.1371/journal.pone.0163105
Degradation Rate of 5-Fluorouracil in Metastatic Colorectal Cancer: A New Predictive Outcome Biomarker?
Abstract
Background: 5-FU based chemotherapy is the most common first line regimen used for metastatic colorectal cancer (mCRC). Identification of predictive markers of response to chemotherapy is a challenging approach for drug selection. The present study analyzes the predictive role of 5-FU degradation rate (5-FUDR) and genetic polymorphisms (MTHFR, TSER, DPYD) on survival.
Materials and methods: Genetic polymorphisms of MTHFR, TSER and DPYD, and the 5-FUDR of homogenous patients with mCRC were retrospectively studied. Genetic markers and the 5-FUDR were correlated with clinical outcome.
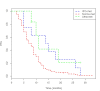
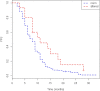
Results: 133 patients affected by mCRC, treated with fluoropyrimidine-based chemotherapy from 2009 to 2014, were evaluated. Patients were classified into three metabolic classes, according to normal distribution of 5-FUDR in more than 1000 patients, as previously published: poor-metabolizer (PM) with 5-FU-DR ≤ 0,85 ng/ml/106 cells/min (8 pts); normal metabolizer with 0,85 < 5-FU-DR < 2,2 ng/ml/106 cells/min (119 pts); ultra-rapid metabolizer (UM) with 5-FU-DR ≥ 2,2 ng/ml/106 cells/min (6 pts). PM and UM groups showed a longer PFS respect to normal metabolizer group (14.5 and 11 months respectively vs 8 months; p = 0.029). A higher G3-4 toxicity rate was observed in PM and UM, respect to normal metabolizer (50% in both PM and UM vs 18%; p = 0.019). No significant associations between genes polymorphisms and outcomes or toxicities were observed.
Conclusion: 5-FUDR seems to be significantly involved in predicting survival of patients who underwent 5-FU based CHT for mCRC. Although our findings require confirmation in large prospective studies, they reinforce the concept that individual genetic variation may allow personalized selection of chemotherapy to optimize clinical outcomes.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Similar articles
-
5-Fluorouracil degradation rate could predict toxicity in stages II-III colorectal cancer patients undergoing adjuvant FOLFOX.Anticancer Drugs. 2017 Mar;28(3):322-326. doi: 10.1097/CAD.0000000000000453. Anticancer Drugs. 2017. PMID: 27845948
-
Evaluation of 5-fluorouracil degradation rate and Pharmacogenetic profiling to predict toxicity following adjuvant Capecitabine.Eur J Clin Pharmacol. 2017 Feb;73(2):157-164. doi: 10.1007/s00228-016-2160-8. Epub 2016 Nov 18. Eur J Clin Pharmacol. 2017. PMID: 27864592
-
Pre-treatment assay of 5-fluorouracil degradation rate (5-FUDR) to improve prediction of 5-fluorouracil toxicity in gastro-esophageal cancer.Oncotarget. 2017 Feb 21;8(8):14050-14057. doi: 10.18632/oncotarget.12571. Oncotarget. 2017. PMID: 27738344 Free PMC article.
-
Basic research supported developments of chemotherapy in nonresectable isolated colorectal liver metastases to a protocol of hepatic artery infusion using mitoxantrone, 5-FU + folinic acid and mitomycin C.Gan To Kagaku Ryoho. 1999 Feb;26(3):269-81. Gan To Kagaku Ryoho. 1999. PMID: 10065089 Review.
-
Thymidylate synthase pharmacogenetics in colorectal cancer.Clin Colorectal Cancer. 2001 Nov;1(3):175-8; discussion 179-81. doi: 10.3816/CCC.2001.n.018. Clin Colorectal Cancer. 2001. PMID: 12450432 Review.
Cited by
-
Recent Updates on Mechanisms of Resistance to 5-Fluorouracil and Reversal Strategies in Colon Cancer Treatment.Biology (Basel). 2021 Aug 31;10(9):854. doi: 10.3390/biology10090854. Biology (Basel). 2021. PMID: 34571731 Free PMC article. Review.
-
Targeted sequencing reveals genetic variants associated with sensitivity of 79 human cancer xenografts to anticancer drugs.Exp Ther Med. 2018 Feb;15(2):1339-1359. doi: 10.3892/etm.2017.5533. Epub 2017 Nov 21. Exp Ther Med. 2018. PMID: 29434720 Free PMC article.
-
Importance of Rare DPYD Genetic Polymorphisms for 5-Fluorouracil Therapy in the Japanese Population.Front Pharmacol. 2022 Jun 15;13:930470. doi: 10.3389/fphar.2022.930470. eCollection 2022. Front Pharmacol. 2022. PMID: 35784703 Free PMC article.
-
Predicting Dihydropyrimidine Dehydrogenase Deficiency and Related 5-Fluorouracil Toxicity: Opportunities and Challenges of DPYD Exon Sequencing and the Role of Phenotyping Assays.Int J Mol Sci. 2022 Nov 11;23(22):13923. doi: 10.3390/ijms232213923. Int J Mol Sci. 2022. PMID: 36430399 Free PMC article.
-
Whole-exome sequencing of 79 xenografts as a potential approach for the identification of genetic variants associated with sensitivity to cytotoxic anticancer drugs.PLoS One. 2020 Sep 28;15(9):e0239614. doi: 10.1371/journal.pone.0239614. eCollection 2020. PLoS One. 2020. PMID: 32986753 Free PMC article.
References
-
- Giachetti S, Perpoint B, Zidani R, Le Bail N, Faggiuolo R, Focan C, et al. Phase III multicenter randomized trial of oxaliplatin added to chronomodulated fluorouracil-leucovorin as first-line treatment of metastatic colorectal cancer. J Clin Oncol. 2000; 18 (1): 136–147. - PubMed
-
- De Gramont A, Figer A, Seymour M, Homerin M, Hmissi A, Cassidy J, et al. Leucovorin and fluorouracil with or without oxaliplatin as firstline treatment in advanced colorectal cancer. J Clin Oncol. 2000; 18 (16): 2938–2947. - PubMed
-
- Cassidy J, Tabernero J, Twelves C, Brunet R, Butts C, Conroy T, et al. XELOX (capecitabine plus oxaliplatin): active first-line therapy for patients with metastatic colorectal cancer. J Clin Oncol. 2004; 22 (11): 2084–2091. - PubMed
-
- Colucci G, Gebbia V, Paoletti G, Giuliani F, Caruso M, Gebbia N, et al. Phase III randomised trial of FOLFIRI versus FOLFOX4 in the treatment of advanced colorectal cancer: a multicenter study of the Gruppo Oncologico Dell’italia Meridonale. J Clin Oncol. 2005; 23 (22): 4866–4875. - PubMed
-
- Longley DB, Harkin DP, Johnston PG. 5-fluorouracil: mechanisms of action and clinical strategies. Nat Rev Cancer. 2003; 3 (5):330–338 - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources