Vimentin, a Novel NF-κB Regulator, Is Required for Meningitic Escherichia coli K1-Induced Pathogen Invasion and PMN Transmigration across the Blood-Brain Barrier
- PMID: 27657497
- PMCID: PMC5033352
- DOI: 10.1371/journal.pone.0162641
Vimentin, a Novel NF-κB Regulator, Is Required for Meningitic Escherichia coli K1-Induced Pathogen Invasion and PMN Transmigration across the Blood-Brain Barrier
Abstract
Background: NF-κB activation, pathogen invasion, polymorphonuclear leukocytes (PMN) transmigration (PMNT) across the blood-brain barrier (BBB) are the pathogenic triad hallmark features of bacterial meningitis, but the mechanisms underlying these events remain largely unknown. Vimentin, which is a novel NF-κB regulator, is the primary receptor for the major Escherichia coli K1 virulence factor IbeA that contributes to the pathogenesis of neonatal bacterial sepsis and meningitis (NSM). We have previously shown that IbeA-induced NF-κB signaling through its primary receptor vimentin as well as its co-receptor PTB-associated splicing factor (PSF) is required for pathogen penetration and leukocyte transmigration across the BBB. This is the first in vivo study to demonstrate how vimentin and related factors contributed to the pathogenic triad of bacterial meningitis.
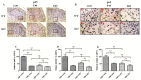
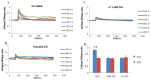
Methodology/principal findings: The role of vimentin in IbeA+ E. coli K1-induced NF-κB activation, pathogen invasion, leukocyte transmigration across the BBB has now been demonstrated by using vimentin knockout (KO) mice. In the in vivo studies presented here, IbeA-induced NF-κB activation, E. coli K1 invasion and polymorphonuclear neutrophil (PMN) transmigration across the BBB were significantly reduced in Vim-/- mice. Decreased neuronal injury in the hippocampal dentate gyrus was observed in Vim-/- mice with meningitis. The major inflammatory regulator α7 nAChR and several signaling molecules contributing to NF-κB activation (p65 and p-CamKII) were significantly reduced in the brain tissues of the Vim-/- mice with E. coli meningitis. Furthermore, Vim KO resulted in significant reduction in neuronal injury and in α7 nAChR-mediated calcium signaling.
Conclusion/significance: Vimentin, a novel NF-κB regulator, plays a detrimental role in the host defense against meningitic infection by modulating the NF-κB signaling pathway to increase pathogen invasion, PMN recruitment, BBB permeability and neuronal inflammation. Our findings provide the first evidence for Vim-dependent mechanisms underlying the pathogenic triad of bacterial meningitis.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




Similar articles
-
Vimentin and PSF act in concert to regulate IbeA+ E. coli K1 induced activation and nuclear translocation of NF-κB in human brain endothelial cells.PLoS One. 2012;7(4):e35862. doi: 10.1371/journal.pone.0035862. Epub 2012 Apr 20. PLoS One. 2012. PMID: 22536447 Free PMC article.
-
Meningitic Escherichia coli K1 penetration and neutrophil transmigration across the blood-brain barrier are modulated by alpha7 nicotinic receptor.PLoS One. 2011;6(9):e25016. doi: 10.1371/journal.pone.0025016. Epub 2011 Sep 22. PLoS One. 2011. PMID: 21966399 Free PMC article.
-
Involvement of IbeA in meningitic Escherichia coli K1-induced polymorphonuclear leukocyte transmigration across brain endothelial cells.Brain Pathol. 2011 Jul;21(4):389-404. doi: 10.1111/j.1750-3639.2010.00463.x. Epub 2010 Dec 13. Brain Pathol. 2011. PMID: 21083634 Free PMC article.
-
Pathogenic Triad in Bacterial Meningitis: Pathogen Invasion, NF-κB Activation, and Leukocyte Transmigration that Occur at the Blood-Brain Barrier.Front Microbiol. 2016 Feb 19;7:148. doi: 10.3389/fmicb.2016.00148. eCollection 2016. Front Microbiol. 2016. PMID: 26925035 Free PMC article. Review.
-
Current concepts on Escherichia coli K1 translocation of the blood-brain barrier.FEMS Immunol Med Microbiol. 2004 Nov 1;42(3):271-9. doi: 10.1016/j.femsim.2004.09.001. FEMS Immunol Med Microbiol. 2004. PMID: 15477040 Review.
Cited by
-
Different Involvement of Vimentin during Invasion by Listeria monocytogenes at the Blood-Brain and the Blood-Cerebrospinal Fluid Barriers In Vitro.Int J Mol Sci. 2022 Oct 26;23(21):12908. doi: 10.3390/ijms232112908. Int J Mol Sci. 2022. PMID: 36361697 Free PMC article.
-
Mechanical and Non-Mechanical Functions of Filamentous and Non-Filamentous Vimentin.Bioessays. 2020 Nov;42(11):e2000078. doi: 10.1002/bies.202000078. Epub 2020 Sep 6. Bioessays. 2020. PMID: 32893352 Free PMC article. Review.
-
Extracellular Vimentin as a Target Against SARS-CoV-2 Host Cell Invasion.Small. 2022 Feb;18(6):e2105640. doi: 10.1002/smll.202105640. Epub 2021 Dec 5. Small. 2022. PMID: 34866333 Free PMC article.
-
Vimentin activation in early apoptotic cancer cells errands survival pathways during DNA damage inducer CPT treatment in colon carcinoma model.Cell Death Dis. 2019 Jun 13;10(6):467. doi: 10.1038/s41419-019-1690-2. Cell Death Dis. 2019. PMID: 31197132 Free PMC article.
-
Host Cell Vimentin Restrains Toxoplasma gondii Invasion and Phosphorylation of Vimentin is Partially Regulated by Interaction with TgROP18.Int J Biol Sci. 2017 Sep 5;13(9):1126-1137. doi: 10.7150/ijbs.21247. eCollection 2017. Int J Biol Sci. 2017. PMID: 29104504 Free PMC article.
References
-
- Huang SH, Stins MF, Kim KS (2000) Bacterial penetration across the blood-brain barrier during the development of neonatal meningitis. Microbes Infect 2: 1237–1244. - PubMed
-
- Kim BY, Kang J, Kim KS (2005) Invasion processes of pathogenic Escherichia coli. Int J Med Microbiol 295: 463–470. - PubMed
-
- Kim KS (2003) Pathogenesis of bacterial meningitis: from bacteraemia to neuronal injury. Nat Rev Neurosci 4: 376–385. - PubMed
-
- Huang SH, Wan ZS, Chen YH, Jong AY, Kim KS (2001) Further characterization of Escherichia coli brain microvascular endothelial cell invasion gene ibeA by deletion, complementation, and protein expression. J Infect Dis 183: 1071–1078. - PubMed
-
- Chen YH, Chen SH, Jong A, Zhou ZY, Li W, Suzuki K, et al. (2002) Enhanced Escherichia coli invasion of human brain microvascular endothelial cells is associated with alternations in cytoskeleton induced by nicotine. Cell Microbiol 4: 503–514. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

