Normal Development and Function of T Cells in Proline Rich 7 (Prr7) Deficient Mice
- PMID: 27657535
- PMCID: PMC5033326
- DOI: 10.1371/journal.pone.0162863
Normal Development and Function of T Cells in Proline Rich 7 (Prr7) Deficient Mice
Abstract
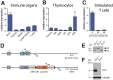
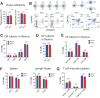
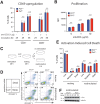
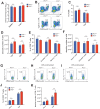
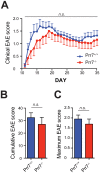
Transmembrane adaptor proteins (TRAPs) are important organisers for the transduction of immunoreceptor-mediated signals. Prr7 is a TRAP that regulates T cell receptor (TCR) signalling and potently induces cell death when overexpressed in human Jurkat T cells. Whether endogenous Prr7 has a similar functional role is currently unknown. To address this issue, we analysed the development and function of the immune system in Prr7 knockout mice. We found that loss of Prr7 partially impairs development of single positive CD4+ T cells in the thymus but has no effect on the development of other T cell subpopulations, B cells, NK cells, or NKT cells. Moreover, Prr7 does not affect the TCR signalling pathway as T cells derived from Prr7 knockout and wild-type animals and stimulated in vitro express the same levels of the activation marker CD69, and retain their ability to proliferate and activate induced cell death programs. Importantly, Prr7 knockout mice retained the capacity to mount a protective immune response when challenged with Listeria monocytogenes infection in vivo. In addition, T cell effector functions (activation, migration, and reactivation) were normal following induction of experimental autoimmune encephalomyelitis (EAE) in Prr7 knockout mice. Collectively, our work shows that loss of Prr7 does not result in a major immune system phenotype and suggests that Prr7 has a dispensable function for TCR signalling.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Similar articles
-
Special AT-rich sequence binding protein 1 is required for maintenance of T cell receptor responsiveness and development of experimental autoimmune encephalomyelitis.Microbiol Immunol. 2018 Apr;62(4):255-268. doi: 10.1111/1348-0421.12579. Epub 2018 Mar 30. Microbiol Immunol. 2018. PMID: 29388727 Free PMC article.
-
A chimeric TCR-beta chain confers increased susceptibility to EAE.Mol Immunol. 2007 Jul;44(14):3473-81. doi: 10.1016/j.molimm.2007.03.015. Epub 2007 May 3. Mol Immunol. 2007. PMID: 17481734
-
Protein kinase B/Akt signals impair Th17 differentiation and support natural regulatory T cell function and induced regulatory T cell formation.J Immunol. 2009 Nov 15;183(10):6124-34. doi: 10.4049/jimmunol.0900246. Epub 2009 Oct 19. J Immunol. 2009. PMID: 19841181
-
Glutamate, T cells and multiple sclerosis.J Neural Transm (Vienna). 2017 Jul;124(7):775-798. doi: 10.1007/s00702-016-1661-z. Epub 2017 Feb 24. J Neural Transm (Vienna). 2017. PMID: 28236206 Review.
-
The molecular mechanisms that control function and death of effector CD4+ T cells.Immunol Res. 2006;36(1-3):275-82. doi: 10.1385/IR:36:1:275. Immunol Res. 2006. PMID: 17337788 Review.
Cited by
-
miR-329- and miR-495-mediated Prr7 down-regulation is required for homeostatic synaptic depression in rat hippocampal neurons.Life Sci Alliance. 2022 Sep 23;5(12):e202201520. doi: 10.26508/lsa.202201520. Life Sci Alliance. 2022. PMID: 36150742 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

