Frontline Dasatinib Treatment in a "Real-Life" Cohort of Patients Older than 65 Years with Chronic Myeloid Leukemia
- PMID: 27659013
- PMCID: PMC5031865
- DOI: 10.1016/j.neo.2016.07.005
Frontline Dasatinib Treatment in a "Real-Life" Cohort of Patients Older than 65 Years with Chronic Myeloid Leukemia
Abstract
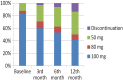
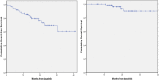
Dasatinib (DAS) has been licensed for the frontline treatment in chronic myeloid leukemia (CML). However, very few data are available regarding its efficacy and toxicity in elderly patients with CML outside clinical trials. To address this issue, we set out a "real-life" cohort of 65 chronic phase CML patients older than 65 years (median age 75.1 years) treated frontline with DAS in 26 Italian centers from June 2012 to June 2015, focusing our attention on toxicity and efficacy data. One third of patients (20/65: 30.7%) had 3 or more comorbidities and required concomitant therapies; according to Sokal classification, 3 patients (4.6%) were low risk, 39 (60.0%) intermediate risk, and 20 (30.8%) high risk, whereas 3 (4.6%) were not classifiable. DAS starting dose was 100 mg once a day in 54 patients (83.0%), whereas 11 patients (17.0%) received less than 100 mg/day. Grade 3/4 hematologic and extrahematologic toxicities were reported in 8 (12.3%) and 12 (18.5%) patients, respectively. Overall, 10 patients (15.4%) permanently discontinued DAS because of toxicities. Pleural effusions (all WHO grades) occurred in 12 patients (18.5%) and in 5 of them occurred during the first 3 months. DAS treatment induced in 60/65 patients (92.3%) a complete cytogenetic response and in 50/65 (76.9%) also a major molecular response. These findings show that DAS might play an important role in the frontline treatment of CML patients >65 years old, proving efficacy and having a favorable safety profile also in elderly subjects with comorbidities.
Copyright © 2016 The Authors. Published by Elsevier Inc. All rights reserved.
Figures
Similar articles
-
Second-line Dasatinib Therapy Improved Compliance and Deep Molecular Responses in Imatinib-intolerant Chronic Myeloid Leukemia Patients.Anticancer Res. 2020 Sep;40(9):5313-5317. doi: 10.21873/anticanres.14538. Anticancer Res. 2020. PMID: 32878823
-
Long term follow-up of frontline Dasatinib in older patients with chronic myeloid leukemia in chronic phase treated outside clinical trials: a real-life cohort observational study.Acta Oncol. 2021 Nov;60(11):1527-1533. doi: 10.1080/0284186X.2021.1971292. Epub 2021 Sep 9. Acta Oncol. 2021. PMID: 34499575
-
Long-term follow-up of lower dose dasatinib (50 mg daily) as frontline therapy in newly diagnosed chronic-phase chronic myeloid leukemia.Cancer. 2020 Jan 1;126(1):67-75. doi: 10.1002/cncr.32504. Epub 2019 Sep 25. Cancer. 2020. PMID: 31553487 Free PMC article. Clinical Trial.
-
Dasatinib: a tyrosine kinase inhibitor for the treatment of chronic myelogenous leukemia and philadelphia chromosome-positive acute lymphoblastic leukemia.Clin Ther. 2007 Nov;29(11):2289-308. doi: 10.1016/j.clinthera.2007.11.005. Clin Ther. 2007. PMID: 18158072 Review.
-
Chronic myeloid leukemia: Second-line drugs of choice.Am J Hematol. 2016 Jan;91(1):67-75. doi: 10.1002/ajh.24247. Am J Hematol. 2016. PMID: 26588811 Review.
Cited by
-
Italian Real-World Analysis of a Tyrosine Kinase Inhibitor Administration as First- or Second-Line of Therapy in Patients with Chronic Myeloid Leukemia.Ther Clin Risk Manag. 2021 Jun 8;17:617-622. doi: 10.2147/TCRM.S309342. eCollection 2021. Ther Clin Risk Manag. 2021. PMID: 34135589 Free PMC article.
-
Contemporary practice patterns of tyrosine kinase inhibitor use among older patients with chronic myeloid leukemia in the United States.Ther Adv Hematol. 2021 Nov 30;12:20406207211043404. doi: 10.1177/20406207211043404. eCollection 2021. Ther Adv Hematol. 2021. PMID: 35154624 Free PMC article.
-
A Pragmatic Approach to Managing Long-Term Adverse Effects in Chronic Myeloid Leukemia Treatment.Curr Hematol Malig Rep. 2023 Aug;18(4):98-104. doi: 10.1007/s11899-023-00698-4. Epub 2023 May 16. Curr Hematol Malig Rep. 2023. PMID: 37191911 Review.
-
Adverse events and dose modifications of tyrosine kinase inhibitors in chronic myelogenous leukemia.Front Oncol. 2022 Oct 6;12:1021662. doi: 10.3389/fonc.2022.1021662. eCollection 2022. Front Oncol. 2022. PMID: 36276124 Free PMC article. Review.
-
CML in the very elderly: the impact of comorbidities and TKI selection in a real-life multicenter study.Ann Hematol. 2024 Sep;103(9):3585-3594. doi: 10.1007/s00277-024-05828-3. Epub 2024 Jun 11. Ann Hematol. 2024. PMID: 38862792 Free PMC article.
References
-
- Druker BJ, Guilhot F, O’Brien SG, Gathmann I, Kantarjian H, Gattermann N, Deininger MW, Silver RT, Goldman JM, Stone RM. Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. N Engl J Med. 2006;355:2408–2417. - PubMed
-
- Branford S, Seymour J, Grigg A, Arthur C, Rudzki Z, Lynch K, Hughes T. BCR-ABL messenger RNA levels continue to decline in patients with chronic phase chronic myeloid leukemia treated with imatinib for more than 5 years and approximately half of all first-line treated patients have stable undetectable BCR-ABL using strict sensitivity criteria. Clin Cancer Res. 2007;13:7080–7086. - PubMed
-
- Stagno F, Stella S, Spitaleri A, Pennisi MS, Di Raimondo F, Vigneri P. Imatinib mesylate in chronic myeloid leukemia: frontline treatment and long-term outcomes. Expert Rev Anticancer Ther. 2016;16(3):273–278. - PubMed
-
- Marin D, Milojkovic D, Olavarria E, Khorashad JS, de Lavallade H, Reid AG, Foroni L, Rezvani K, Bua M, Dazzi F. European LeukemiaNet criteria for failure or suboptimal response reliably identify patients with CML in early chronic phase treated with imatinib whose eventual outcome is poor. Blood. 2008;112:4437–4444. - PMC - PubMed
-
- Castagnetti F, Gugliotta G, Breccia M, Stagno F, Iurlo A, Albano F, Abruzzese E, Martino B, Levato L, Intermesoli T. Long-term outcome of chronic myeloid leukemia patients treated frontline with imatinib. Leukemia. 2015;29(9):1823–1831. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical