Central Control Circuit for Context-Dependent Micturition
- PMID: 27662084
- PMCID: PMC6217838
- DOI: 10.1016/j.cell.2016.08.073
Central Control Circuit for Context-Dependent Micturition
Abstract
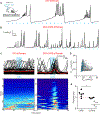
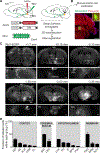
Urine release (micturition) serves an essential physiological function as well as a critical role in social communication in many animals. Here, we show a combined effect of olfaction and social hierarchy on micturition patterns in adult male mice, confirming the existence of a micturition control center that integrates pro- and anti-micturition cues. Furthermore, we demonstrate that a cluster of neurons expressing corticotropin-releasing hormone (Crh) in the pontine micturition center (PMC) is electrophysiologically distinct from their Crh-negative neighbors and sends glutamatergic projections to the spinal cord. The activity of PMC Crh-expressing neurons correlates with and is sufficient to drive bladder contraction, and when silenced impairs micturition behavior. These neurons receive convergent input from widespread higher brain areas that are capable of carrying diverse pro- and anti-micturition signals, and whose activity modulates hierarchy-dependent micturition. Taken together, our results indicate that PMC Crh-expressing neurons are likely the integration center for context-dependent micturition behavior.
Keywords: Barrington's nucleus; bladder; corticotropin-releasing hormone; medial pre-optic area; micturition; pons.
Copyright © 2016 Elsevier Inc. All rights reserved.
Conflict of interest statement
COMPETING FINANCIAL INTERESTS
The authors declare no competing financial interests.
Figures





References
-
- Bacchini A, Gaetani E, and Cavaggioni A (1992). Pheromone binding proteins of the mouse,Mus musculus. Experientia 48, 419–421. - PubMed
-
- Barrington FJF (1925). The effect of lesions of the hind- and mid-brain on micturition in the cat. Exp. Physiol 15, 81–102.
-
- Betts CD, Kapoor R, and Fowler CJ (1993). Pontine Pathology and Voiding Dysfunction. Br. J. Urol 72, 100–102. - PubMed
-
- Beynon RJ, Veggerby C, Payne CE, Robertson DHL, Gaskell SJ, Humphries RE, and Hurst JL (2002). Polymorphism in major urinary proteins: molecular heterogeneity in a wild mouse population. J. Chem. Ecol 28, 1429–1446. - PubMed
-
- Blok BFM, and Holstege G (1998). The central nervous system control of micturition in cats and humans. Behav. Brain Res 92, 119–125. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

