A Novel Conserved Domain Mediates Dimerization of Protein Kinase D (PKD) Isoforms: DIMERIZATION IS ESSENTIAL FOR PKD-DEPENDENT REGULATION OF SECRETION AND INNATE IMMUNITY
- PMID: 27662904
- PMCID: PMC5095407
- DOI: 10.1074/jbc.M116.735399
A Novel Conserved Domain Mediates Dimerization of Protein Kinase D (PKD) Isoforms: DIMERIZATION IS ESSENTIAL FOR PKD-DEPENDENT REGULATION OF SECRETION AND INNATE IMMUNITY
Abstract
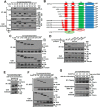
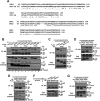
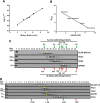
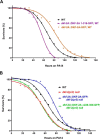
Protein kinase D (PKD) isoforms are protein kinase C effectors in signaling pathways regulated by diacylglycerol. Important physiological processes (including secretion, immune responses, motility, and transcription) are placed under diacylglycerol control by the distinctive substrate specificity and subcellular distribution of PKDs. Potentially, broadly co-expressed PKD polypeptides may interact to generate homo- or heteromultimeric regulatory complexes. However, the frequency, molecular basis, regulatory significance, and physiological relevance of stable PKD-PKD interactions are largely unknown. Here, we demonstrate that mammalian PKDs 1-3 and the prototypical Caenorhabditis elegans PKD, DKF-2A, are exclusively (homo- or hetero-) dimers in cell extracts and intact cells. We discovered and characterized a novel, highly conserved N-terminal domain, comprising 92 amino acids, which mediates dimerization of PKD1, PKD2, and PKD3 monomers. A similar domain directs DKF-2A homodimerization. Dimerization occurred independently of properties of the regulatory and kinase domains of PKDs. Disruption of PKD dimerization abrogates secretion of PAUF, a protein carried in small trans-Golgi network-derived vesicles. In addition, disruption of DKF-2A homodimerization in C. elegans intestine impaired and degraded the immune defense of the intact animal against an ingested bacterial pathogen. Finally, dimerization was indispensable for the strong, dominant negative effect of catalytically inactive PKDs. Overall, the structural integrity and function of the novel dimerization domain are essential for PKD-mediated regulation of a key aspect of cell physiology, secretion, and innate immunity in vivo.
Keywords: Caenorhabditis elegans (C. elegans); heterodimer; homodimer; innate immunity; native Mr; oligomerization domain; protein kinase D (PKD); secretion; signal transduction.
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures



References
-
- Ellwanger K., and Hausser A. (2013) Physiological functions of protein kinase D in vivo. IUBMB Life 65, 98–107 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

