Polo-like Kinase 1 Regulates Vimentin Phosphorylation at Ser-56 and Contraction in Smooth Muscle
- PMID: 27662907
- PMCID: PMC5095422
- DOI: 10.1074/jbc.M116.749341
Polo-like Kinase 1 Regulates Vimentin Phosphorylation at Ser-56 and Contraction in Smooth Muscle
Abstract
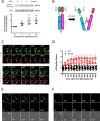
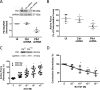
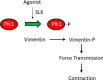
Polo-like kinase 1 (Plk1) is a serine/threonine-protein kinase that has been implicated in mitosis, cytokinesis, and smooth muscle cell proliferation. The role of Plk1 in smooth muscle contraction has not been investigated. Here, stimulation with acetylcholine induced Plk1 phosphorylation at Thr-210 (an indication of Plk1 activation) in smooth muscle. Contractile stimulation also activated Plk1 in live smooth muscle cells as evidenced by changes in fluorescence resonance energy transfer signal of a Plk1 sensor. Moreover, knockdown of Plk1 in smooth muscle attenuated force development. Smooth muscle conditional knock-out of Plk1 also diminished contraction of mouse tracheal rings. Plk1 knockdown inhibited acetylcholine-induced vimentin phosphorylation at Ser-56 without affecting myosin light chain phosphorylation. Expression of T210A Plk1 inhibited the agonist-induced vimentin phosphorylation at Ser-56 and contraction in smooth muscle. However, myosin light chain phosphorylation was not affected by T210A Plk1. Ste20-like kinase (SLK) is a serine/threonine-protein kinase that has been implicated in spindle orientation and microtubule organization during mitosis. In this study knockdown of SLK inhibited Plk1 phosphorylation at Thr-210 and activation. Finally, asthma is characterized by airway hyperresponsiveness, which largely stems from airway smooth muscle hyperreactivity. Here, smooth muscle conditional knock-out of Plk1 attenuated airway resistance and airway smooth muscle hyperreactivity in a murine model of asthma. Taken together, these findings suggest that Plk1 regulates smooth muscle contraction by modulating vimentin phosphorylation at Ser-56. Plk1 activation is regulated by SLK during contractile activation. Plk1 contributes to the pathogenesis of asthma.
Keywords: cytoskeleton; excitation-contraction coupling (E-C coupling); intermediate filament; phosphorylation; signal transduction; smooth muscle.
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures




Similar articles
-
Ste20-like Kinase-mediated Control of Actin Polymerization Is a New Mechanism for Thin Filament-associated Regulation of Airway Smooth Muscle Contraction.Am J Respir Cell Mol Biol. 2020 May;62(5):645-656. doi: 10.1165/rcmb.2019-0310OC. Am J Respir Cell Mol Biol. 2020. PMID: 31913659 Free PMC article.
-
Plk1 Mediates Paxillin Phosphorylation (Ser-272), Centrosome Maturation, and Airway Smooth Muscle Layer Thickening in Allergic Asthma.Sci Rep. 2019 May 17;9(1):7555. doi: 10.1038/s41598-019-43927-8. Sci Rep. 2019. PMID: 31101859 Free PMC article.
-
MicroRNA miR-509 Regulates ERK1/2, the Vimentin Network, and Focal Adhesions by Targeting Plk1.Sci Rep. 2018 Aug 22;8(1):12635. doi: 10.1038/s41598-018-30895-8. Sci Rep. 2018. PMID: 30135525 Free PMC article.
-
Playing polo during mitosis: PLK1 takes the lead.Oncogene. 2017 Aug 24;36(34):4819-4827. doi: 10.1038/onc.2017.113. Epub 2017 Apr 24. Oncogene. 2017. PMID: 28436952 Review.
-
Polo-like kinase 1 (PLK1) signaling in cancer and beyond.Biochem Pharmacol. 2021 Nov;193:114747. doi: 10.1016/j.bcp.2021.114747. Epub 2021 Aug 26. Biochem Pharmacol. 2021. PMID: 34454931 Review.
Cited by
-
Nestin Modulates Airway Smooth Muscle Cell Migration by Affecting Spatial Rearrangement of Vimentin Network and Focal Adhesion Assembly.Cells. 2022 Sep 29;11(19):3047. doi: 10.3390/cells11193047. Cells. 2022. PMID: 36231009 Free PMC article.
-
Role and regulation of Abelson tyrosine kinase in Crk-associated substrate/profilin-1 interaction and airway smooth muscle contraction.Respir Res. 2018 Jan 5;19(1):4. doi: 10.1186/s12931-017-0709-4. Respir Res. 2018. PMID: 29304860 Free PMC article.
-
Inhibiting Airway Smooth Muscle Contraction Using Pitavastatin: A Role for the Mevalonate Pathway in Regulating Cytoskeletal Proteins.Front Pharmacol. 2020 May 6;11:469. doi: 10.3389/fphar.2020.00469. eCollection 2020. Front Pharmacol. 2020. PMID: 32435188 Free PMC article.
-
PLK1 Mediates the Proliferation and Contraction of Airway Smooth Muscle Cells and Has a Role in T2-High Asthma with Neutrophilic Inflammation Model.J Inflamm Res. 2025 Mar 25;18:4381-4394. doi: 10.2147/JIR.S501645. eCollection 2025. J Inflamm Res. 2025. PMID: 40162075 Free PMC article.
-
Current Understanding of Asthma Pathogenesis and Biomarkers.Cells. 2022 Sep 5;11(17):2764. doi: 10.3390/cells11172764. Cells. 2022. PMID: 36078171 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

