Disinhibition of neurons of the nucleus of solitary tract that project to the superior salivatory nucleus causes choroidal vasodilation: Implications for mechanisms underlying choroidal baroregulation
- PMID: 27663135
- PMCID: PMC5117681
- DOI: 10.1016/j.neulet.2016.09.029
Disinhibition of neurons of the nucleus of solitary tract that project to the superior salivatory nucleus causes choroidal vasodilation: Implications for mechanisms underlying choroidal baroregulation
Abstract
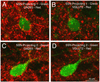
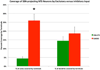
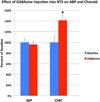
Preganglionic neurons in the superior salivatory nucleus (SSN) that mediate parasympathetic vasodilation of choroidal blood vessels receive a major excitatory input from the baroresponsive part of the nucleus of the solitary tract (NTS). This input appears likely to mediate choroidal vasodilation during systemic hypotension, which prevents decreases in choroidal blood flow (ChBF) due to reduced perfusion pressure. It is uncertain, however, how low blood pressure signals to NTS from the aortic depressor nerve (ADN), which fires at a low rate during systemic hypotension, could yield increased firing in the NTS output to SSN. The simplest hypothesis is that SSN-projecting NTS neurons are under the inhibitory control of ADN-receptive GABAergic NTS neurons. As part of evaluating this hypothesis, we assessed if SSN-projecting NTS neurons, in fact, receive prominent inhibitory input and if blocking GABAergic modulation of them increases ChBF. We found that SSN-projecting NTS neuronal perikarya identified by retrograde labeling are densely coated with GABAergic terminals, but lightly coated with excitatory terminals. We also found that, infusion of the GABA-A receptor antagonist GABAzine into NTS increased ChBF. Our results are consistent with the possibility that low blood pressure signals from the ADN produce vasodilation in choroid by causing diminished activity in ADN-receptive NTS neurons that tonically suppress SSN-projecting NTS neurons.
Keywords: Autonomic; Choroidal blood flow; Disinhibition; GABA; Nucleus of solitary tract; Parasympathetic; Superior salivatory nucleus.
Copyright © 2016 Elsevier Ireland Ltd. All rights reserved.
Figures

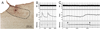
Similar articles
-
Role of the superior salivatory nucleus in parasympathetic control of choroidal blood flow and in maintenance of retinal health.Exp Eye Res. 2021 May;206:108541. doi: 10.1016/j.exer.2021.108541. Epub 2021 Mar 16. Exp Eye Res. 2021. PMID: 33736985 Free PMC article.
-
Stimulation of Baroresponsive Parts of the Nucleus of the Solitary Tract Produces Nitric Oxide-mediated Choroidal Vasodilation in Rat Eye.Front Neuroanat. 2016 Oct 7;10:94. doi: 10.3389/fnana.2016.00094. eCollection 2016. Front Neuroanat. 2016. PMID: 27774055 Free PMC article.
-
Projections from the hypothalamic paraventricular nucleus and the nucleus of the solitary tract to prechoroidal neurons in the superior salivatory nucleus: Pathways controlling rodent choroidal blood flow.Brain Res. 2010 Oct 28;1358:123-39. doi: 10.1016/j.brainres.2010.08.065. Epub 2010 Aug 27. Brain Res. 2010. PMID: 20801105 Free PMC article.
-
The identification and neurochemical characterization of central neurons that target parasympathetic preganglionic neurons involved in the regulation of choroidal blood flow in the rat eye using pseudorabies virus, immunolabeling and conventional pathway tracing methods.Front Neuroanat. 2015 Jun 2;9:65. doi: 10.3389/fnana.2015.00065. eCollection 2015. Front Neuroanat. 2015. PMID: 26082687 Free PMC article.
-
Plasticity of GABAergic mechanisms within the nucleus of the solitary tract in hypertension.Hypertension. 2010 Feb;55(2):201-6. doi: 10.1161/HYPERTENSIONAHA.109.146407. Epub 2010 Jan 4. Hypertension. 2010. PMID: 20048192 Free PMC article. Review. No abstract available.
Cited by
-
Differential Functional Changes in Visual Performance during Acute Exposure to Microgravity Analogue and Their Potential Links with Spaceflight-Associated Neuro-Ocular Syndrome.Diagnostics (Basel). 2024 Aug 30;14(17):1918. doi: 10.3390/diagnostics14171918. Diagnostics (Basel). 2024. PMID: 39272703 Free PMC article.
-
Role of the superior salivatory nucleus in parasympathetic control of choroidal blood flow and in maintenance of retinal health.Exp Eye Res. 2021 May;206:108541. doi: 10.1016/j.exer.2021.108541. Epub 2021 Mar 16. Exp Eye Res. 2021. PMID: 33736985 Free PMC article.
-
Stimulation of Baroresponsive Parts of the Nucleus of the Solitary Tract Produces Nitric Oxide-mediated Choroidal Vasodilation in Rat Eye.Front Neuroanat. 2016 Oct 7;10:94. doi: 10.3389/fnana.2016.00094. eCollection 2016. Front Neuroanat. 2016. PMID: 27774055 Free PMC article.
-
Defective Choroidal Blood Flow Baroregulation and Retinal Dysfunction and Pathology Following Sympathetic Denervation of Choroid.Invest Ophthalmol Vis Sci. 2018 Oct 1;59(12):5032-5044. doi: 10.1167/iovs.18-24954. Invest Ophthalmol Vis Sci. 2018. PMID: 30326072 Free PMC article.
-
Vagal Nerve Stimulation Attenuates Ischemia-Reperfusion Induced Retina Dysfunction in Acute Ocular Hypertension.Front Neurosci. 2019 Feb 11;13:87. doi: 10.3389/fnins.2019.00087. eCollection 2019. Front Neurosci. 2019. PMID: 30804746 Free PMC article.
References
-
- Agassandian K, Fazan VP, Adanina V, Talman WT. Direct projections from the cardiovascular nucleus tractus solitarii to pontine preganglionic parasympathetic neurons: a link to cerebrovascular regulation. J. Comp. Neurol. 2002;452:242–254. - PubMed
-
- Bevan MD, Crossman AR, Bolam JP. Neurons projecting from the entopeduncular nucleus to the thalamus receive convergent synaptic inputs from the subthalamic nucleus and the neostriatum in the rat. Brain Res. 1994;659:99–109. - PubMed
-
- Ciriello J. Brainstem projections of aortic baroreceptor afferent fibers in the rat. Neurosci. Lett. 1983;36:37–42. - PubMed
-
- Crill WE, Reis DJ. Distribution of carotid sinus and depressor nerves in cat brain stem. Am. J. Physiol. 1968;214:269–276. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

