F-actin rearrangement is regulated by mTORC2/Akt/Girdin in mouse fertilized eggs
- PMID: 27666957
- PMCID: PMC6496183
- DOI: 10.1111/cpr.12285
F-actin rearrangement is regulated by mTORC2/Akt/Girdin in mouse fertilized eggs
Abstract
In mouse fertilized eggs, correct assembly and distribution of the actin cytoskeleton are intimately related to cleavage in early-stage embryos. However, in mouse fertilized eggs, mechanisms and involved factors responsible for regulating the actin cytoskeleton are poorly defined. In this study, mTORC2, PKB/Akt and Girdin were found to modulate division of mouse fertilized eggs by regulating distribution of the actin cytoskeleton. RNA interference (RNAi)-mediated depletion of mTORC2, Akt1 or Girdin disrupted F-actin rearrangement and strongly inhibited egg development. PKB/Akt has been proven to be a downstream target of the mTORC2 signalling pathway. Girdin, a newly found actin cross-linker, has been proven to be a downstream target of the Akt signalling pathway. Furthermore, phosphorylation of both Akt1 and girdin was affected by knockdown of mTORC2. Akt1 positively regulated development of the mouse fertilized eggs by girdin-mediated F-actin rearrangement. Thus it seems that girdin could be a downstream target of the Akt1-mediated signalling pathway. Collectively, this study aimed to prove participation of mTORC2/Akt in F-actin assembly in early-stage cleavage of mouse fertilized eggs via the function of girdin.
Objectives: In mouse fertilized eggs, the proper assembly and distribution of actin cytoskeleton is intimately related with the cleavage of early-stage embryo. However, in mammals, especially in mouse fertilized eggs, the mechanisms and involved factors responsible for regulating the actin cytoskeleton are poorly defined. The aim of this study was to determine the role of mTORC2,PKB/Akt and Girdin in early development of fertilized mouse eggs, via regulating the distribution of actin cytoskeleton.
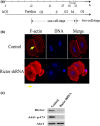
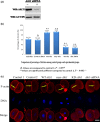
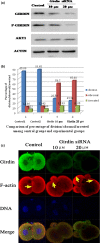
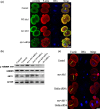
Materials and methods: Changes of F-actin after treatting with mTORC2 shRNA, Akt siRNA or Girdin siRNA were observed by Immunofluorescence staining and laser-scanning confocal microscopy. Levels of phosphorylated Girdin at Se1417 were detected by Western immunoblotting. Percentages of cells undergoing division were determined by counting, using a dissecting microscope.
Results: RNA interference (RNAi)-mediated depletion of mTORC2, Akt1 or Girdin disrupts F-actin rearrangement, and remarkably inhibited the development of mouse-fertilized eggs. PKB/Akt has been proved to be a downstream target of the mTORC2 signaling pathway. Girdin, the newly found actin-cross linker, has been proved to be a downstream target of the Akt signaling pathway. Furthermore phosphorylation of both Akt1 and Girdin were affected by knockdown of mTORC2. Akt1 positively regulates the development of mouse-fertilized eggs by Girdin mediated F-actin rearrangement. Girdin could be a downstream target of the Akt1-mediated signaling pathway.
Conclusions: Collectively, this study aimed to prove the participation of mTORC2/Akt in F-actin assembling in early-stage cleavage of mouse fertilized eggs via the function of Girdin.
© 2016 The Authors Cell Proliferation Published by John Wiley & Sons Ltd.
Conflict of interest statement
All authors have contributed significantly and are in agreement with the content of the manuscript. There is no conflict of interest.
Figures


Similar articles
-
[PKB/Akt regulates the aggregation of actin by Girdin in mouse fertilized eggs].Sheng Wu Gong Cheng Xue Bao. 2016 Sep 25;32(9):1204-1211. doi: 10.13345/j.cjb.160024. Sheng Wu Gong Cheng Xue Bao. 2016. PMID: 29022321 Chinese.
-
Endothelial Cell mTOR Complex-2 Regulates Sprouting Angiogenesis.PLoS One. 2015 Aug 21;10(8):e0135245. doi: 10.1371/journal.pone.0135245. eCollection 2015. PLoS One. 2015. PMID: 26295809 Free PMC article.
-
Influence of proline-rich inositol polyphosphate 5-phosphatase, on early development of fertilized mouse eggs, via inhibition of phosphorylation of Akt.Cell Prolif. 2011 Apr;44(2):156-65. doi: 10.1111/j.1365-2184.2011.00743.x. Cell Prolif. 2011. PMID: 21401757 Free PMC article.
-
Girdin, a novel actin-binding protein, and its family of proteins possess versatile functions in the Akt and Wnt signaling pathways.Ann N Y Acad Sci. 2006 Nov;1086:169-84. doi: 10.1196/annals.1377.016. Ann N Y Acad Sci. 2006. PMID: 17185515 Review.
-
Phosphorylation of Akt at the C-terminal tail triggers Akt activation.Cell Cycle. 2014;13(14):2162-4. doi: 10.4161/cc.29584. Epub 2014 Jun 16. Cell Cycle. 2014. PMID: 24933731 Free PMC article. Review.
Cited by
-
WAVE2 Regulates Actin-Dependent Processes Induced by the B Cell Antigen Receptor and Integrins.Cells. 2023 Nov 25;12(23):2704. doi: 10.3390/cells12232704. Cells. 2023. PMID: 38067132 Free PMC article.
-
Construction of Chitosan-Zn-Based Electrochemical Biosensing Platform for Rapid and Accurate Assay of Actin.Sensors (Basel). 2018 Jun 7;18(6):1865. doi: 10.3390/s18061865. Sensors (Basel). 2018. PMID: 29875352 Free PMC article.
-
High Dosages of Equine Chorionic Gonadotropin Exert Adverse Effects on the Developmental Competence of IVF-Derived Mouse Embryos and Cause Oxidative Stress-Induced Aneuploidy.Front Cell Dev Biol. 2021 Feb 9;8:609290. doi: 10.3389/fcell.2020.609290. eCollection 2020. Front Cell Dev Biol. 2021. PMID: 33634101 Free PMC article.
-
mTORC2 interactome and localization determine aggressiveness of high-grade glioma cells through association with gelsolin.Sci Rep. 2023 Apr 29;13(1):7037. doi: 10.1038/s41598-023-33872-y. Sci Rep. 2023. PMID: 37120454 Free PMC article.
-
Genome sequencing reveals CCDC88A variants in malformations of cortical development and immune dysfunction.Hum Mol Genet. 2025 Jul 20;34(15):1294-1312. doi: 10.1093/hmg/ddaf081. Hum Mol Genet. 2025. PMID: 40401444 Free PMC article.
References
-
- Pickering SJ, Johnson MH, Braude PR, Houliston E. Cytoskeletal organization in fresh, aged and spontaneously activated human oocytes. Hum Reprod. 1998;3:8–89. - PubMed
-
- Zhu ZY, Chen DY, Li JS, et al. Rotation of meiotic spindle is controlled by microfilaments in mouse oocytes. Biol Reprod. 2003;68:943–946. - PubMed
-
- Jacinto E, Loewith R, Schmidt A, et al. Mammalian TOR complex 2 controls the actin cytoskeleton and is rapamycin insensitive. Nat Cell Biol. 2004;6:1122–1128. - PubMed
-
- Sarbassov DD, Ali SM, Kim DH, et al. Rictor, a novel binding partner of mTOR, defines a rapamycin‐insensitive and raptor‐independent pathway that regulates the cytoskeleton. Curr Biol. 2004;14:1296–1302. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

