Modelling TFE renal cell carcinoma in mice reveals a critical role of WNT signaling
- PMID: 27668431
- PMCID: PMC5036965
- DOI: 10.7554/eLife.17047
Modelling TFE renal cell carcinoma in mice reveals a critical role of WNT signaling
Abstract
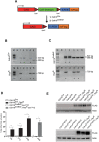
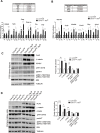
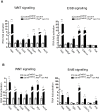
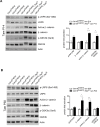
TFE-fusion renal cell carcinomas (TFE-fusion RCCs) are caused by chromosomal translocations that lead to overexpression of the TFEB and TFE3 genes (Kauffman et al., 2014). The mechanisms leading to kidney tumor development remain uncharacterized and effective therapies are yet to be identified. Hence, the need to model these diseases in an experimental animal system (Kauffman et al., 2014). Here, we show that kidney-specific TFEB overexpression in transgenic mice, resulted in renal clear cells, multi-layered basement membranes, severe cystic pathology, and ultimately papillary carcinomas with hepatic metastases. These features closely recapitulate those observed in both TFEB- and TFE3-mediated human kidney tumors. Analysis of kidney samples revealed transcriptional induction and enhanced signaling of the WNT β-catenin pathway. WNT signaling inhibitors normalized the proliferation rate of primary kidney cells and significantly rescued the disease phenotype in vivo. These data shed new light on the mechanisms underlying TFE-fusion RCCs and suggest a possible therapeutic strategy based on the inhibition of the WNT pathway.
Keywords: TFEB; WNT pathway; cancer biology; human biology; medicine; mouse; renal cell carcinoma.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures











References
-
- Amin MB, Amin MB, Tamboli P, Javidan J, Stricker H, Venturina MD-P, Deshpande A, Menon M, de-Peralta Venturina M. Prognostic impact of histologic subtyping of adult renal epithelial neoplasms: an experience of 405 cases. The American Journal of Surgical Pathology. 2002;26:281–291. doi: 10.1097/00000478-200203000-00001. - DOI - PubMed
-
- Argani P, Antonescu CR, Illei PB, Lui MY, Timmons CF, Newbury R, Reuter VE, Garvin AJ, Perez-Atayde AR, Fletcher JA, Beckwith JB, Bridge JA, Ladanyi M. Primary renal neoplasms with the ASPL-TFE3 gene fusion of alveolar soft part sarcoma: a distinctive tumor entity previously included among renal cell carcinomas of children and adolescents. The American Journal of Pathology. 2001;159:179–192. doi: 10.1016/S0002-9440(10)61684-7. - DOI - PMC - PubMed
-
- Argani P, Hicks J, De Marzo AM, Albadine R, Illei PB, Ladanyi M, Reuter VE, Netto GJ. Xp11 translocation renal cell carcinoma (RCC): extended immunohistochemical profile emphasizing novel RCC markers. The American Journal of Surgical Pathology. 2010;34:1295–1303. doi: 10.1097/PAS.0b013e3181e8ce5b. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

