Autophagy in adhesion and migration
- PMID: 27672021
- PMCID: PMC5087656
- DOI: 10.1242/jcs.188490
Autophagy in adhesion and migration
Abstract
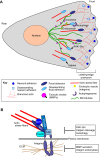
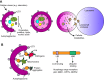
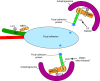
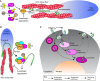
Autophagy, a pathway for lysosomal-mediated cellular degradation, has recently been described as a regulator of cell migration. Although the molecular mechanisms underlying autophagy-dependent motility are only beginning to emerge, new work demonstrates that selective autophagy mediated by the autophagy cargo receptor, NBR1, specifically promotes the dynamic turnover of integrin-based focal adhesion sites during motility. Here, we discuss the detailed mechanisms through which NBR1-dependent selective autophagy supports focal adhesion remodeling, and we describe the interconnections between this pathway and other established regulators of focal adhesion turnover, such as microtubules. We also highlight studies that examine the contribution of autophagy to selective degradation of proteins that mediate cellular tension and to integrin trafficking; these findings hint at further roles for autophagy in supporting adhesion and migration. Given the recently appreciated importance of selective autophagy in diverse cellular processes, we propose that further investigation into autophagy-mediated focal adhesion turnover will not only shed light onto how focal adhesions are regulated but will also unveil new mechanisms regulating selective autophagy.
Keywords: Autophagy; Focal adhesion; Migration; NBR1.
© 2016. Published by The Company of Biologists Ltd.
Conflict of interest statement
The authors declare no competing or financial interests.
Figures
Similar articles
-
NBR1-dependent selective autophagy is required for efficient cell-matrix adhesion site disassembly.Autophagy. 2016 Oct 2;12(10):1958-1959. doi: 10.1080/15548627.2016.1212789. Epub 2016 Aug 2. Autophagy. 2016. PMID: 27484104 Free PMC article.
-
NBR1 enables autophagy-dependent focal adhesion turnover.J Cell Biol. 2016 Feb 29;212(5):577-90. doi: 10.1083/jcb.201503075. Epub 2016 Feb 22. J Cell Biol. 2016. PMID: 26903539 Free PMC article.
-
Autophagy promotes cell motility by driving focal adhesion turnover.Autophagy. 2016 Oct 2;12(10):1685-1686. doi: 10.1080/15548627.2016.1212791. Epub 2016 Aug 2. Autophagy. 2016. PMID: 27483986 Free PMC article.
-
The Interconnections between Autophagy and Integrin-Mediated Cell Adhesion.J Mol Biol. 2017 Feb 17;429(4):515-530. doi: 10.1016/j.jmb.2016.11.027. Epub 2016 Dec 6. J Mol Biol. 2017. PMID: 27932295 Free PMC article. Review.
-
Integrin and microtubule crosstalk in the regulation of cellular processes.Cell Mol Life Sci. 2018 Nov;75(22):4177-4185. doi: 10.1007/s00018-018-2913-x. Epub 2018 Sep 11. Cell Mol Life Sci. 2018. PMID: 30206641 Free PMC article. Review.
Cited by
-
Cell adhesion molecule IGPR-1 activates AMPK connecting cell adhesion to autophagy.J Biol Chem. 2020 Dec 4;295(49):16691-16699. doi: 10.1074/jbc.RA120.014790. Epub 2020 Sep 25. J Biol Chem. 2020. PMID: 32978258 Free PMC article.
-
Epithelial-mesenchymal plasticity in cancer: signaling pathways and therapeutic targets.MedComm (2020). 2024 Aug 1;5(8):e659. doi: 10.1002/mco2.659. eCollection 2024 Aug. MedComm (2020). 2024. PMID: 39092293 Free PMC article. Review.
-
Glioblastoma: new therapeutic strategies to address cellular and genomic complexity.Oncotarget. 2017 Dec 20;9(10):9540-9554. doi: 10.18632/oncotarget.23476. eCollection 2018 Feb 6. Oncotarget. 2017. PMID: 29507709 Free PMC article. Review.
-
Strain-induced mechanoresponse depends on cell contractility and BAG3-mediated autophagy.Mol Biol Cell. 2021 Oct 1;32(20):ar9. doi: 10.1091/mbc.E21-05-0254. Epub 2021 Aug 11. Mol Biol Cell. 2021. PMID: 34379447 Free PMC article.
-
Dysregulated autophagy-related genes in septic cardiomyopathy: Comprehensive bioinformatics analysis based on the human transcriptomes and experimental validation.Front Cardiovasc Med. 2022 Aug 2;9:923066. doi: 10.3389/fcvm.2022.923066. eCollection 2022. Front Cardiovasc Med. 2022. PMID: 35983185 Free PMC article.
References
-
- Belaid A., Cerezo M., Chargui A., Corcelle-Termeau E., Pedeutour F., Giuliano S., Ilie M., Rubera I., Tauc M., Barale S. et al. (2013). Autophagy plays a critical role in the degradation of active RHOA, the control of cell cytokinesis, and genomic stability. Cancer Res. 73, 4311-4322. 10.1158/0008-5472.CAN-12-4142 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

