Incidence, Severity, and Outcomes of AKI Associated with Dual Renin-Angiotensin System Blockade
- PMID: 27679519
- PMCID: PMC5108193
- DOI: 10.2215/CJN.03470316
Incidence, Severity, and Outcomes of AKI Associated with Dual Renin-Angiotensin System Blockade
Abstract
Background and objectives: The benefit of dual blockade of the renin-angiotensin system is limited by adverse effects. We performed a secondary analysis of the Veterans Affairs Nephropathy in Diabetes (VA NEPHRON-D) Study to describe the effect of increased intensity of renin-angiotensin system blockade on the incidence, risk factors, and outcomes of AKI.
Design, setting, participants, & measurements: In the VA NEPHRON-D Study, we randomized 1148 veterans receiving outpatient care with type 2 diabetes mellitus, eGFR of between 30 and 89.9 ml/min per 1.73 m2, and urinary albumin excretion of at least 300 μg/mg creatinine (or a urinary total protein of at least 0.5 mg/mg creatinine) to either combination therapy with losartan and lisinopril or monotherapy with losartan. We identified hospitalized AKI events and their outcomes during a median follow-up of 2.2 years through systematic reporting of serious adverse events.
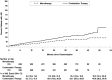
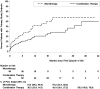
Results: The incidence of AKI was 12.2 (95% confidence interval, 10.5 to 14.0) versus 6.7 (95% confidence interval, 5.6 to 8.2) per 100 patient-years in the combination arm versus monotherapy arms (P<0.001). Individuals with AKI were more likely to develop the primary study end point of death, ESRD, or decline in kidney function (hazard ratio, 1.78; 95% confidence interval, 1.34 to 2.26; P<0.001). Patients with AKI in the combination arm had greater recovery of kidney function (75.9% versus 66.3%; P=0.04), lower 30-day mortality (4.7% versus 15.0%; P<0.01), and lower hazard for development of the primary study end point (hazard ratio, 0.60; 95% confidence interval, 0.37 to 0.98).
Conclusions: Dual renin-angiotensin system blockade was associated with an increased risk of AKI compared with monotherapy, but AKI in the setting of monotherapy was associated with lower rates of recovery of kidney function, higher mortality, and higher risk of progression of kidney disease.
Keywords: Albumins; Ambulatory Care; Arm; Diabetes Mellitus, Type 2; Follow-Up Studies; Humans; Incidence; Kidney Failure, Chronic; Lisinopril; Losartan; Nephrons; Renin-Angiotensin System; Veterans; acute kidney injury; angiotensin converting enzyme inhibitor; angiotensin receptor blocker; chronic kidney disease; creatinine; diabetes mellitus; diabetic nephropathy; glomerular filtration rate; proteinuria; renin angiotensin system; risk factors.
Copyright © 2016 by the American Society of Nephrology.
Figures
References
-
- de Zeeuw D, Remuzzi G, Parving HH, Keane WF, Zhang Z, Shahinfar S, Snapinn S, Cooper ME, Mitch WE, Brenner BM: Proteinuria, a target for renoprotection in patients with type 2 diabetic nephropathy: Lessons from RENAAL. Kidney Int 65: 2309–2320, 2004 - PubMed
-
- Kidney Disease: Improving Global Outcomes (KDIGO) Work Group : KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl 3: 1–150, 2013 - PubMed
-
- Lewis EJ, Hunsicker LG, Bain RP, Rohde RD; The Collaborative Study Group : The effect of angiotensin-converting-enzyme inhibition on diabetic nephropathy. N Engl J Med 329: 1456–1462, 1993 - PubMed
-
- Lewis EJ, Hunsicker LG, Clarke WR, Berl T, Pohl MA, Lewis JB, Ritz E, Atkins RC, Rohde R, Raz I; Collaborative Study Group : Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med 345: 851–860, 2001 - PubMed
-
- Brenner BM, Cooper ME, de Zeeuw D, Keane WF, Mitch WE, Parving HH, Remuzzi G, Snapinn SM, Zhang Z, Shahinfar S; RENAAL Study Investigators : Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med 345: 861–869, 2001 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

