Antibacterial effects of volatiles produced by Bacillus strain D13 against Xanthomonas oryzae pv. oryzae
- PMID: 27682316
- PMCID: PMC6637998
- DOI: 10.1111/mpp.12494
Antibacterial effects of volatiles produced by Bacillus strain D13 against Xanthomonas oryzae pv. oryzae
Abstract
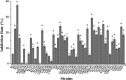
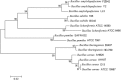
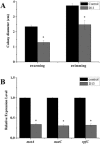
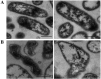
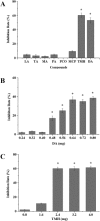
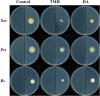
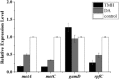
Recent investigations have demonstrated that bacteria employ the volatile compounds they produce during interactions with other organisms, such as plants, fungi, nematodes and bacteria. However, studies focused on the antibacterial activity of plant growth-promoting rhizobacteria (PGPR) volatiles against bacterial phytopathogens are still rare. In this study, Bacillus strain D13, which is antagonistic to Xanthomonas oryzae pv. oryzae (Xoo), was isolated and screened. Volatile compounds emitted from strain D13 reduced the colony diameter and cell motility of Xoo cultured in divided Petri plates. Transmission electron micrograph analysis showed concentration in cytoplasm and altered surface morphology in the majority of Xanthomonas cells after co-cultivation with strain D13. Transcriptional expression of virulence-associated genes in Xoo was repressed. Based on gas chromatography/mass spectrometry (GC/MS) analysis, 12 volatile compounds specifically produced by strain D13 were identified. Among them, decyl alcohol and 3,5,5-trimethylhexanol inhibited the growth of Xoo at minimum inhibitory amounts of 0.48 and 2.4 mg, respectively. Furthermore, transcriptional expression of virulence-associated genes was also repressed by decyl alcohol and 3,5,5-trimethylhexanol. These results are useful for a better understanding of the biocontrol mechanisms of Bacillus.
Keywords: Bacillus; antibacterial activity; volatile compounds, Xanthomonas oryzae pv. oryzae.
© 2016 BSPP AND JOHN WILEY & SONS LTD.
Figures

Similar articles
-
Broad-spectrum antagonistic potential of Bacillus spp. volatiles against Rhizoctonia solani and Xanthomonas oryzae pv. oryzae.Physiol Plant. 2023 Nov-Dec;175(6):e14087. doi: 10.1111/ppl.14087. Physiol Plant. 2023. PMID: 38148207
-
A thiadiazole reduces the virulence of Xanthomonas oryzae pv. oryzae by inhibiting the histidine utilization pathway and quorum sensing.Mol Plant Pathol. 2018 Jan;19(1):116-128. doi: 10.1111/mpp.12503. Epub 2016 Dec 27. Mol Plant Pathol. 2018. PMID: 27756112 Free PMC article.
-
The Antibiosis Action and Rice-Induced Resistance, Mediated by a Lipopeptide from Bacillus amyloliquefaciens B014, in Controlling Rice Disease Caused by Xanthomonas oryzae pv. oryzae.J Microbiol Biotechnol. 2016 Apr 28;26(4):748-56. doi: 10.4014/jmb.1510.10072. J Microbiol Biotechnol. 2016. PMID: 26718470
-
Bacillus species: factories of plant protective volatile organic compounds.J Appl Microbiol. 2023 Mar 1;134(3):lxad037. doi: 10.1093/jambio/lxad037. J Appl Microbiol. 2023. PMID: 36822621 Review.
-
Volatile Nitrogenous Compounds from Bacteria: Source of Novel Bioactive Compounds.Chem Biodivers. 2021 Nov;18(11):e2100549. doi: 10.1002/cbdv.202100549. Epub 2021 Oct 13. Chem Biodivers. 2021. PMID: 34643327 Review.
Cited by
-
Antifungal Effects of Volatiles Produced by Bacillus subtilis Against Alternaria solani in Potato.Front Microbiol. 2020 Jun 17;11:1196. doi: 10.3389/fmicb.2020.01196. eCollection 2020. Front Microbiol. 2020. PMID: 32625175 Free PMC article.
-
A plant growth-promoting bacteria Priestia megaterium JR48 induces plant resistance to the crucifer black rot via a salicylic acid-dependent signaling pathway.Front Plant Sci. 2022 Nov 10;13:1046181. doi: 10.3389/fpls.2022.1046181. eCollection 2022. Front Plant Sci. 2022. PMID: 36438094 Free PMC article.
-
Harnessing microbial volatiles to replace pesticides and fertilizers.Microb Biotechnol. 2020 Sep;13(5):1366-1376. doi: 10.1111/1751-7915.13645. Epub 2020 Aug 7. Microb Biotechnol. 2020. PMID: 32767638 Free PMC article. Review.
-
Volatile compounds reveal age: a study of volatile organic compounds released by Chrysomya rufifacies immatures.Int J Legal Med. 2021 May;135(3):967-977. doi: 10.1007/s00414-020-02471-1. Epub 2020 Nov 23. Int J Legal Med. 2021. PMID: 33230566
-
Microbial Volatile Organic Compounds: An Alternative for Chemical Fertilizers in Sustainable Agriculture Development.Microorganisms. 2022 Dec 22;11(1):42. doi: 10.3390/microorganisms11010042. Microorganisms. 2022. PMID: 36677334 Free PMC article. Review.
References
-
- Altman, J. and Lawlor, S. (1966) The effects of some chlorinated hydrocarbons on certain soil bacteria. J. Appl. Bacteriol. 29, 260–265. - PubMed
-
- Chatterjee, S. , Newman, K.L. and Lindow, S.E. (2008) Cell‐to‐cell signaling in Xylella fastidiosa suppresses movement and xylem vessel colonization in grape. Mol. Plant–Microbe Interact. 21, 1309–1315. - PubMed
-
- Corcuff, R. , Mercier, J. , Tweddell, R. and Arul, J. (2011) Effect of water activity on the production of volatile organic compounds by Muscodor albus and their effect on three pathogens in stored potato. Fungal Biol. 115, 220–227. - PubMed
-
- Dainty, R. , Edwards, R. and Hibbard, C. (1985) Time course of volatile compound formation during refrigerated storage of naturally contaminated beef in air. J. Appl. Bacteriol. 59, 303–309. - PubMed
-
- Domsch, K. (1959) Die wirkung von bodenfungiciden III. Quantitative varenderungen der bodenflora. Z. Pflanzenkr. Pflanzenschutz, 66, 17–26.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

