Long Term Ex Vivo Culture and Live Imaging of Drosophila Larval Imaginal Discs
- PMID: 27685172
- PMCID: PMC5042436
- DOI: 10.1371/journal.pone.0163744
Long Term Ex Vivo Culture and Live Imaging of Drosophila Larval Imaginal Discs
Abstract
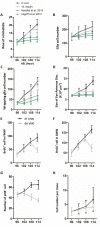
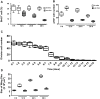
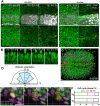
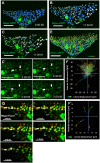
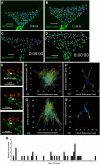
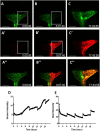
Continuous imaging of live tissues provides clear temporal sequence of biological events. The Drosophila imaginal discs have been popular experimental subjects for the study of a wide variety of biological phenomena, but long term culture that allows normal development has not been satisfactory. Here we report a culture method that can sustain normal development for 18 hours and allows live imaging. The method is validated in multiple discs and for cell proliferation, differentiation and migration. However, it does not support disc growth and cannot support cell proliferation for more than 7 to 12 hr. We monitored the cellular behavior of retinal basal glia in the developing eye disc and found that distinct glia type has distinct properties of proliferation and migration. The live imaging provided direct proof that wrapping glia differentiated from existing glia after migrating to the anterior front, and unexpectedly found that they undergo endoreplication before wrapping axons, and their nuclei migrate up and down along the axons. UV-induced specific labeling of a single carpet glia also showed that the two carpet glia membrane do not overlap and suggests a tiling or repulsion mechanism between the two cells. These findings demonstrated the usefulness of an ex vivo culture method and live imaging.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
Long-term Live Imaging of Drosophila Eye Disc.J Vis Exp. 2017 May 6;(123):55748. doi: 10.3791/55748. J Vis Exp. 2017. PMID: 28518091 Free PMC article.
-
Early lineage segregation of the retinal basal glia in the Drosophila eye disc.Sci Rep. 2020 Oct 28;10(1):18522. doi: 10.1038/s41598-020-75581-w. Sci Rep. 2020. PMID: 33116242 Free PMC article.
-
Oxygenation and adenosine deaminase support growth and proliferation of ex vivo cultured Drosophila wing imaginal discs.Development. 2017 Jul 1;144(13):2529-2538. doi: 10.1242/dev.147538. Epub 2017 May 19. Development. 2017. PMID: 28526754
-
Cultivation and Live Imaging of Drosophila Imaginal Discs.Methods Mol Biol. 2016;1478:203-213. doi: 10.1007/978-1-4939-6371-3_11. Methods Mol Biol. 2016. PMID: 27730583 Review.
-
Stem cell plasticity in mammals and transdetermination in Drosophila: common themes?Stem Cells. 2000;18(6):409-14. doi: 10.1634/stemcells.18-6-409. Stem Cells. 2000. PMID: 11072028 Review.
Cited by
-
Expressional Profiling of Carpet Glia in the Developing Drosophila Eye Reveals Its Molecular Signature of Morphology Regulators.Front Neurosci. 2019 Mar 29;13:244. doi: 10.3389/fnins.2019.00244. eCollection 2019. Front Neurosci. 2019. PMID: 30983950 Free PMC article.
-
Long-term Live Imaging of Drosophila Eye Disc.J Vis Exp. 2017 May 6;(123):55748. doi: 10.3791/55748. J Vis Exp. 2017. PMID: 28518091 Free PMC article.
-
Ex Vivo Calcium Imaging for Visualizing Brain Responses to Endocrine Signaling in Drosophila.J Vis Exp. 2018 Jun 2;(136):57701. doi: 10.3791/57701. J Vis Exp. 2018. PMID: 29912190 Free PMC article.
-
Cultivation and Live Imaging of Drosophila Imaginal Discs.Methods Mol Biol. 2022;2540:317-334. doi: 10.1007/978-1-0716-2541-5_16. Methods Mol Biol. 2022. PMID: 35980586
-
The fly eye: Through the looking glass.Dev Dyn. 2018 Jan;247(1):111-123. doi: 10.1002/dvdy.24585. Epub 2017 Oct 23. Dev Dyn. 2018. PMID: 28856763 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

