The eIF2A knockout mouse
- PMID: 27686860
- PMCID: PMC5134716
- DOI: 10.1080/15384101.2016.1237324
The eIF2A knockout mouse
Abstract
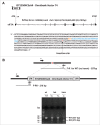
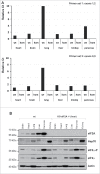
Eukaryotic initiation factor 2A (eIF2A) is a 65-kDa protein that was first identified in the early 1970s as a factor capable of stimulating initiator methionyl-tRNAi (Met-tRNAMeti) binding to 40S ribosomal subunits in vitro. However, in contrast to the eIF2, which stimulates Met-tRNAMeti binding to 40S ribosomal subunits in a GTP-dependent manner, eIF2A didn't reveal any GTP-dependence, but instead was found to direct binding of the Met-tRNAMeti to 40S ribosomal subunits in a codon-dependent manner. eIF2A appears to be highly conserved across eukaryotic species, suggesting conservation of function in evolution. The yeast Saccharomyces cerevisae eIF2A null mutant revealed no apparent phenotype, however, it was found that in yeast eIF2A functions as a suppressor of internal ribosome entry site (IRES)-mediated translation. It was thus suggested that eIF2A my act by impinging on the expression of specific mRNAs. Subsequent studies in mammalian cell systems implicated eIF2A in non-canonical (non-AUG-dependent) translation initiation events involving near cognate UUG and CUG codons. Yet, the role of eIF2A in cellular functions remains largely enigmatic. As a first step toward characterization of the eIF2A function in mammalian systems in vivo, we have obtained homozygous eIF2A-total knockout (KO) mice, in which a gene trap cassette was inserted between eIF2A exons 1 and 2 disrupting expression of all exons downstream of the insertion. The KO mice strain is viable and to date displays no apparent phenotype. We believe that the eIF2A KO mice strain will serve as a valuable tool for researchers studying non-canonical initiation of translation in vivo.
Keywords: alternative initiation pathways; eIF2A; initiation of translation; knockout mouse; non-AUG dependent initiation.
Figures

References
-
- Jackson RJ, Hellen CU, Pestova TV. The mechanism of eukaryotic translation initiation and principles of its regulation. Nat Rev Mol Cell Biol 2010; 11:113-27; PMID:20094052; http://dx.doi.org/10.1038/nrm2838 - DOI - PMC - PubMed
-
- Hinnebusch AG. The scanning mechanism of eukaryotic translation initiation. Annu Rev Biochem 2014; 83:779-812; PMID:24499181; http://dx.doi.org/10.1146/annurev-biochem-060713-035802 - DOI - PubMed
-
- Merrick WC, Anderson WF. Purification and characterization of homogeneous protein synthesis initiation factor M1 from rabbit reticulocytes. J Biol Chem 1975; 250:1197-206; PMID:1112800 - PubMed
-
- Adams SL, Safer B, Anderson WF, Merrick WC. Eukaryotic initiation complex formation. Evidence for two distinct pathways. J Biol Chem 1975; 250:9083-89; PMID:1194278 - PubMed
-
- Benne R, Brown-Luedi ML, Hershey JW. Protein synthesis initiation factors from rabbit reticulocytes: purification, characterization, and radiochemical labeling. Methods Enzymol 1979; 60:15-35; PMID:459896; http://dx.doi.org/10.1016/S0076-6879(79)60005-8 - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous
