Decreased Expression of SRSF2 Splicing Factor Inhibits Apoptotic Pathways in Renal Cancer
- PMID: 27690003
- PMCID: PMC5085631
- DOI: 10.3390/ijms17101598
Decreased Expression of SRSF2 Splicing Factor Inhibits Apoptotic Pathways in Renal Cancer
Erratum in
-
Correction: Kędzierska, H., et al. Decreased Expression of SRSF2 Splicing Factor Inhibits Apoptotic Pathways in Renal Cancer. Int. J. Mol. Sci. 2016, 17, 1598.Int J Mol Sci. 2020 Mar 30;21(7):2380. doi: 10.3390/ijms21072380. Int J Mol Sci. 2020. PMID: 32235585 Free PMC article.
Abstract
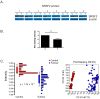
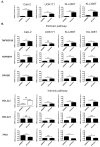
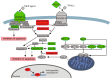
Serine and arginine rich splicing factor 2(SRSF2) belongs to the serine/arginine (SR)-rich family of proteins that regulate alternative splicing. Previous studies suggested that SRSF2 can contribute to carcinogenic processes. Clear cell renal cell carcinoma (ccRCC) is the most common subtype of kidney cancer, highly aggressive and difficult to treat, mainly due to resistance to apoptosis. In this study we hypothesized that SRSF2 contributes to the regulation of apoptosis in ccRCC. Using tissue samples obtained from ccRCC patients, as well as independent validation on The Cancer Genome Atlas (TCGA) data, we demonstrate for the first time that expression of SRSF2 is decreased in ccRCC tumours when compared to non-tumorous control tissues. Furthermore, by employing a panel of ccRCC-derived cell lines with silenced SRSF2 expression and qPCR arrays we show that SRSF2 contributes not only to splicing patterns but also to expression of multiple apoptotic genes, including new SRSF2 targets: DIABLO, BIRC5/survivin, TRAIL, BIM, MCL1, TNFRSF9, TNFRSF1B, CRADD, BCL2L2, BCL2A1, and TP53. We also identified a new splice variant of CFLAR, an inhibitor of caspase activity. These changes culminate in diminished caspase-9 activity and inhibition of apoptosis. In summary, we show for the first time that decreased expression of SRSF2 in ccRCC contributes to protection of cancer cells viability.
Keywords: CFLAR; SRSF2; TCGA; alternative splicing; apoptosis; caspase-9; ccRCC; mRNA; renal cancer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
microRNA-mediated regulation of splicing factors SRSF1, SRSF2 and hnRNP A1 in context of their alternatively spliced 3'UTRs.Exp Cell Res. 2018 Feb 15;363(2):208-217. doi: 10.1016/j.yexcr.2018.01.009. Epub 2018 Jan 10. Exp Cell Res. 2018. PMID: 29331391
-
Serine-arginine splicing factor 2 promotes oesophageal cancer progression by regulating alternative splicing of interferon regulatory factor 3.RNA Biol. 2023 Jan;20(1):359-367. doi: 10.1080/15476286.2023.2223939. RNA Biol. 2023. PMID: 37335045 Free PMC article.
-
SRSF1 and SRSF2 synergistically regulate Bim expression to mediate glucocorticoid-induced apoptosis in osteoblasts.Biochem Biophys Res Commun. 2025 Jul 12;770:152015. doi: 10.1016/j.bbrc.2025.152015. Epub 2025 May 14. Biochem Biophys Res Commun. 2025. PMID: 40381239
-
Splicing factors of SR and hnRNP families as regulators of apoptosis in cancer.Cancer Lett. 2017 Jun 28;396:53-65. doi: 10.1016/j.canlet.2017.03.013. Epub 2017 Mar 14. Cancer Lett. 2017. PMID: 28315432 Review.
-
Splicing factor SRSF2-centric gene regulation.Int J Biol Sci. 2021 Apr 16;17(7):1708-1715. doi: 10.7150/ijbs.58888. eCollection 2021. Int J Biol Sci. 2021. PMID: 33994855 Free PMC article. Review.
Cited by
-
Correction: Kędzierska, H., et al. Decreased Expression of SRSF2 Splicing Factor Inhibits Apoptotic Pathways in Renal Cancer. Int. J. Mol. Sci. 2016, 17, 1598.Int J Mol Sci. 2020 Mar 30;21(7):2380. doi: 10.3390/ijms21072380. Int J Mol Sci. 2020. PMID: 32235585 Free PMC article.
-
BCL-w: apoptotic and non-apoptotic role in health and disease.Cell Death Dis. 2020 Apr 21;11(4):260. doi: 10.1038/s41419-020-2417-0. Cell Death Dis. 2020. PMID: 32317622 Free PMC article. Review.
-
Environmental perturbations lead to extensive directional shifts in RNA processing.PLoS Genet. 2017 Oct 12;13(10):e1006995. doi: 10.1371/journal.pgen.1006995. eCollection 2017 Oct. PLoS Genet. 2017. PMID: 29023442 Free PMC article.
-
Tipping the balance of cell death: alternative splicing as a source of MCL-1S in cancer.Cell Death Dis. 2024 Dec 18;15(12):917. doi: 10.1038/s41419-024-07307-z. Cell Death Dis. 2024. PMID: 39695189 Free PMC article. Review.
-
Survival-Associated Alternative Splicing Events in Pan-Renal Cell Carcinoma.Front Oncol. 2019 Nov 27;9:1317. doi: 10.3389/fonc.2019.01317. eCollection 2019. Front Oncol. 2019. PMID: 31850211 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

