Intracellular d-Serine Accumulation Promotes Genetic Diversity via Modulated Induction of RecA in Enterohemorrhagic Escherichia coli
- PMID: 27698085
- PMCID: PMC5116935
- DOI: 10.1128/JB.00548-16
Intracellular d-Serine Accumulation Promotes Genetic Diversity via Modulated Induction of RecA in Enterohemorrhagic Escherichia coli
Abstract
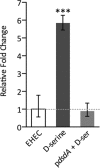
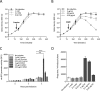
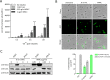
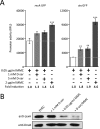
We recently discovered that exposure of enterohemorrhagic Escherichia coli (EHEC) to d-serine resulted in accumulation of this unusual amino acid, induction of the SOS regulon, and downregulation of the type III secretion system that is essential for efficient colonization of the host. Here, we have investigated the physiological relevance of this elevated SOS response, which is of particular interest given the presence of Stx toxin-carrying lysogenic prophages on the EHEC chromosome that are activated during the SOS response. We found that RecA elevation in response to d-serine, while being significant, was heterogeneous and not capable of activating stx expression or stx phage transduction to a nonlysogenic recipient. This "SOS-like response" was, however, capable of increasing the mutation frequency associated with low-level RecA activity, thus promoting genetic diversity. Furthermore, this response was entirely dependent on RecA and enhanced in the presence of a DNA-damaging agent, indicating a functional SOS response, but did not result in observable cleavage of the LexA repressor alone, indicating a controlled mechanism of induction. This work demonstrates that environmental factors not usually associated with DNA damage are capable of promoting an SOS-like response. We propose that this modulated induction of RecA allows EHEC to adapt to environmental insults such as d-serine while avoiding unwanted phage-induced lysis.
Importance: The SOS response is a global stress network that is triggered by the presence of DNA damage due to breakage or stalled replication forks. Activation of the SOS response can trigger the replication of lytic bacteriophages and promote genetic diversification through error-prone polymerases. We have demonstrated that the host-associated metabolite d-serine contributes to Escherichia coli niche specification and accumulates inside cells that cannot catabolize it. This results in a modulated activation of the SOS antirepressor RecA that is insufficient to trigger lytic bacteriophage but capable of increasing the SOS-associated mutation frequency. These findings describe how relevant signals not normally associated with DNA damage can hijack the SOS response, promoting diversity as E. coli strains adapt while avoiding unwanted phage lysis.
Copyright © 2016, American Society for Microbiology. All Rights Reserved.
Figures

References
-
- Fernández De Henestrosa AR, Ogi T, Aoyagi S, Chafin D, Hayes JJ, Ohmori H, Woodgate R. 2000. Identification of additional genes belonging to the LexA regulon in Escherichia coli. Mol Microbiol 35:1560–1572. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

