Dysregulation of Autophagy Contributes to Anal Carcinogenesis
- PMID: 27706233
- PMCID: PMC5051741
- DOI: 10.1371/journal.pone.0164273
Dysregulation of Autophagy Contributes to Anal Carcinogenesis
Abstract
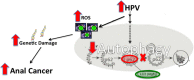
Introduction: Autophagy is an intracellular catabolic process that removes and recycles unnecessary/dysfunctional cellular components, contributing to cellular health and survival. Autophagy is a highly regulated cellular process that responds to several intracellular signals, many of which are deregulated by human papillomavirus (HPV) infection through the expression of HPV-encoded oncoproteins. This adaptive inhibitory response helps prevent viral clearance. A strong correlation remains between HPV infection and the development of squamous cell carcinoma (SCC) of the anus, particularly in HIV positive and other immunosuppressed patients. We hypothesize that autophagy is inhibited by HPV-encoded oncoproteins thereby promoting anal carcinogenesis (Fig 1).
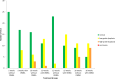
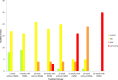
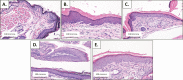
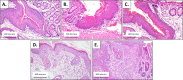
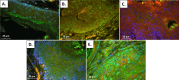
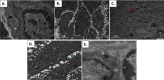
Materials and methods: HPV16 transgenic mice (K14E6/E7) and non-transgenic mice (FVB/N), both of which do not spontaneously develop anal tumors, were treated topically with the chemical carcinogen, 7,12-Dimethylbenz[a]anthracene (DMBA), to induce anal cancer. The anuses at different time points of treatment (5, 10, 15 and 20 weeks) were analyzed using immunofluorescence (IF) for two key autophagy marker proteins (LC3β and p62) in addition to histological grading. The anuses from the K14E6/E7 mice were also analyzed for visual evidence of autophagic activity by electron microscopy (EM). To see if there was a correlation to humans, archival anal specimens were assessed histologically for grade of dysplasia and then analyzed for LC3β and p62 protein content. To more directly examine the effect of autophagic inhibition on anal carcinogenesis, nontransgenic mice that do not develop anal cancer with DMBA treatment were treated with a known pharmacologic inhibitor of autophagy, chloroquine, and examined for tumor development and analyzed by IF for autophagic proteins.
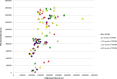
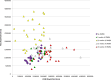
Results: Histologically, we observed the progression of normal anoderm to invasive SCC with DMBA treatment in K14E6/E7 mice but not in nontransgenic, syngeneic FVB/N background control mice. With the development of low-grade dysplasia in the K14E6/E7 mice, there was an increase in both punctate LC3β and p62 expression while EM revealed increased autophagosomes without evidence of autophagolysosomes. These observations are consistent with autophagy being inhibited at a later stage in the autophagic process. In contrast, in high-grade dysplasia and SCC in the DMBA-treated K14E6/E7 mice, there were decreased levels of p62 with a continued increase in punctate LC3β expression by IF, while autophagolysosomes were seen on EM, consistent with the process of autophagy proceeded to completion. Similar findings, including histological grade dependent changes in LC3β and p62 expression, were noted with human samples upon analysis of IF. Finally, with pharmacologic inhibition of autophagy in DMBA-treated, nontrangenic FVB/N mice, there was a significant increase in anal cancer development similar to that observed in DMBA- treated K14E6/E7 mice.
Conclusion: Autophagic dysregulation is noted early on in HPV-associated anal carcinogenesis (low-grade dysplasia), with normalization of the autophagic process arising in late stages of HPV-associated anal carcinogenesis (high-grade dysplasia and invasive carcinoma).
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures





References
-
- SEER Cancer Statistics Factsheets: Anal cancer. National Cancer Institute; Bethesda, MD, Available: http://seer.cancer.gov/statfacts/html/anus.html. Updated November 18, 2015. Accessed 4 April 2016.
-
- Baricevic I, He X, Chakrabarty B, Oliver AW, Bailey C, Summers J, et al. High-sensitvity human papilloma virus genotyping reveals near universal positivity in anal sqaumous cell carcinoma: different implications for vaccine prevention and prognosis. Eur J Cancer. 2015, 51 (6): 776–85. 10.1016/j.ejca.2015.01.058 - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

