Inducible Expression of the De-Novo Designed Antimicrobial Peptide SP1-1 in Tomato Confers Resistance to Xanthomonas campestris pv. vesicatoria
- PMID: 27706237
- PMCID: PMC5051901
- DOI: 10.1371/journal.pone.0164097
Inducible Expression of the De-Novo Designed Antimicrobial Peptide SP1-1 in Tomato Confers Resistance to Xanthomonas campestris pv. vesicatoria
Abstract
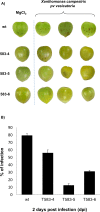
Antimicrobial peptides (AMPs) are small peptides with less than 50 amino acids and are part of the innate immune response in almost all organisms, including bacteria, vertebrates, invertebrates and plants. AMPs are active against a broad-spectrum of pathogens. The inducible expression of AMPs in plants is a promising approach to combat plant pathogens with minimal negative side effects, such as phytotoxicity or infertility. In this study, inducible expression of the de-novo designed AMP SP1-1 in Micro Tom tomato protected tomato fruits against bacterial spot disease caused by Xanthomonas campestris pv. vesicatoria. The peptide SP1-1 was targeted to the apoplast which is the primary infection site for plant pathogens, by fusing SP1-1 peptide to the signal peptide RsAFP1 of radish (Raphanus sativus). The pathogen inducibility of the expression was enabled by using an optimized inducible 4XW2/4XS promoter. As a result, the tomato fruits of independently generated SP1-1 transgenic lines were significantly more resistant to X. campestris pv. vesicatoria than WT tomato fruits. In transgenic lines, bacterial infection was reduced up to 65% in comparison to the infection of WT plants. Our study demonstrates that the combination of the 4XW2/4XS cis-element from parsley with the synthetic antimicrobial peptide SP1-1 is a good alternative to protect tomato fruits against infections with X. campestris pv. vesicatoria.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
Identification and expression profiling of tomato genes differentially regulated during a resistance response to Xanthomonas campestris pv. vesicatoria.Mol Plant Microbe Interact. 2004 Nov;17(11):1212-22. doi: 10.1094/MPMI.2004.17.11.1212. Mol Plant Microbe Interact. 2004. PMID: 15553246
-
Genetic mapping and functional analysis of the tomato Bs4 locus governing recognition of the Xanthomonas campestris pv. vesicatoria AvrBs4 protein.Mol Plant Microbe Interact. 2001 May;14(5):629-38. doi: 10.1094/MPMI.2001.14.5.629. Mol Plant Microbe Interact. 2001. PMID: 11332727
-
Xanthomonas campestris pv. vesicatoria effector AvrBsT induces cell death in pepper, but suppresses defense responses in tomato.Mol Plant Microbe Interact. 2010 Aug;23(8):1069-82. doi: 10.1094/MPMI-23-8-1069. Mol Plant Microbe Interact. 2010. PMID: 20615117
-
Advances and Challenges in Bacterial Spot Resistance Breeding in Tomato (Solanum lycopersicum L.).Int J Mol Sci. 2020 Mar 3;21(5):1734. doi: 10.3390/ijms21051734. Int J Mol Sci. 2020. PMID: 32138355 Free PMC article. Review.
-
Genomic approaches in Xanthomonas campestris pv. vesicatoria allow fishing for virulence genes.J Biotechnol. 2003 Dec 19;106(2-3):203-14. doi: 10.1016/j.jbiotec.2003.07.012. J Biotechnol. 2003. PMID: 14651862 Review.
Cited by
-
Recent advances in developing disease resistance in plants.F1000Res. 2019 Nov 19;8:F1000 Faculty Rev-1934. doi: 10.12688/f1000research.20179.1. eCollection 2019. F1000Res. 2019. PMID: 31807281 Free PMC article. Review.
-
Rapid Colorimetric Detection of Bacterial Species through the Capture of Gold Nanoparticles by Chimeric Phages.ACS Nano. 2019 Feb 26;13(2):1244-1252. doi: 10.1021/acsnano.8b06395. Epub 2019 Jan 2. ACS Nano. 2019. PMID: 30586498 Free PMC article.
-
Advances of Peptides for Plant Immunity.Plants (Basel). 2025 Aug 7;14(15):2452. doi: 10.3390/plants14152452. Plants (Basel). 2025. PMID: 40805801 Free PMC article. Review.
-
Fusion of a chitin-binding domain to an antibacterial peptide to enhance resistance to Fusarium solani in tobacco (Nicotiana tabacum).3 Biotech. 2018 Sep;8(9):391. doi: 10.1007/s13205-018-1416-7. Epub 2018 Aug 28. 3 Biotech. 2018. PMID: 30175028 Free PMC article.
-
Antimicrobial Peptides: The Production of Novel Peptide-Based Therapeutics in Plant Systems.Life (Basel). 2023 Sep 7;13(9):1875. doi: 10.3390/life13091875. Life (Basel). 2023. PMID: 37763279 Free PMC article. Review.
References
-
- Nan YH, Park KH, Park Y, Jeon YJ, Kim Y, Park IS, et al. (2009) Investigating the effects of positive charge and hydrophobicity on the cell selectivity, mechanism of action and anti-inflammatory activity of a Trp-rich antimicrobial peptide indolicidin. FEMS Microbiol Lett 292: 134–140. 10.1111/j.1574-6968.2008.01484.x - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

