Syncytial Mutations Do Not Impair the Specificity of Entry and Spread of a Glycoprotein D Receptor-Retargeted Herpes Simplex Virus
- PMID: 27707922
- PMCID: PMC5126368
- DOI: 10.1128/JVI.01456-16
Syncytial Mutations Do Not Impair the Specificity of Entry and Spread of a Glycoprotein D Receptor-Retargeted Herpes Simplex Virus
Abstract
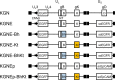
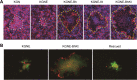
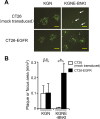
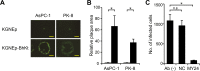
Membrane fusion, which is the key process for both initial cell entry and subsequent lateral spread of herpes simplex virus (HSV), requires the four envelope glycoproteins gB, gD, gH, and gL. Syncytial mutations, predominantly mapped to the gB and gK genes, confer hyperfusogenicity on HSV and cause multinucleated giant cells, termed syncytia. Here we asked whether interaction of gD with a cognate entry receptor remains indispensable for initiating membrane fusion of syncytial strains. To address this question, we took advantage of mutant viruses whose viral entry into cells relies on the uniquely specific interaction of an engineered gD with epidermal growth factor receptor (EGFR). We introduced selected syncytial mutations into gB and/or gK of the EGFR-retargeted HSV and found that these mutations, especially when combined, enabled formation of extensive syncytia by human cancer cell lines that express the target receptor; these syncytia were substantially larger than the plaques formed by the parental retargeted HSV strain. We assessed the EGFR dependence of entry and spread separately by using direct entry and infectious center assays, respectively, and we found that the syncytial mutations did not override the receptor specificity of the retargeted viruses at either stage. We discuss the implications of these results for the development of more effective targeted oncolytic HSV vectors.
Importance: Herpes simplex virus (HSV) is investigated not only as a human pathogen but also as a promising agent for oncolytic virotherapy. We previously showed that both the initial entry and subsequent lateral spread of HSV can be retargeted to cells expressing tumor-associated antigens by single-chain antibodies fused to a receptor-binding-deficient envelope glycoprotein D (gD). Here we introduced syncytial mutations into the gB and/or gK gene of gD-retargeted HSVs to determine whether viral tropism remained dependent on the interaction of gD with the target receptor. Entry and spread profiles of the recombinant viruses indicated that gD retargeting does not abolish the hyperfusogenic activity of syncytial mutations and that these mutations do not eliminate the dependence of HSV entry and spread on a specific gD-receptor interaction. These observations suggest that syncytial mutations may be valuable for increasing the tumor-specific spreading of retargeted oncolytic HSV vectors.
Copyright © 2016, American Society for Microbiology. All Rights Reserved.
Figures


Similar articles
-
Antibody Screening System Using a Herpes Simplex Virus (HSV)-Based Probe To Identify a Novel Target for Receptor-Retargeted Oncolytic HSVs.J Virol. 2021 Apr 12;95(9):e01766-20. doi: 10.1128/JVI.01766-20. Print 2021 Apr 12. J Virol. 2021. PMID: 33627393 Free PMC article.
-
Development of an oncolytic HSV vector fully retargeted specifically to cellular EpCAM for virus entry and cell-to-cell spread.Gene Ther. 2016 Jun;23(6):479-88. doi: 10.1038/gt.2016.17. Epub 2016 Feb 23. Gene Ther. 2016. PMID: 26905369
-
Dual Ligand Insertion in gB and gD of Oncolytic Herpes Simplex Viruses for Retargeting to a Producer Vero Cell Line and to Cancer Cells.J Virol. 2018 Feb 26;92(6):e02122-17. doi: 10.1128/JVI.02122-17. Print 2018 Mar 15. J Virol. 2018. PMID: 29263257 Free PMC article.
-
Two Sides to Every Story: Herpes Simplex Type-1 Viral Glycoproteins gB, gD, gH/gL, gK, and Cellular Receptors Function as Key Players in Membrane Fusion.Viruses. 2021 Sep 16;13(9):1849. doi: 10.3390/v13091849. Viruses. 2021. PMID: 34578430 Free PMC article. Review.
-
Retargeting Strategies for Oncolytic Herpes Simplex Viruses.Viruses. 2016 Feb 26;8(3):63. doi: 10.3390/v8030063. Viruses. 2016. PMID: 26927159 Free PMC article. Review.
Cited by
-
Entry of Alphaherpesviruses.Curr Issues Mol Biol. 2021;41:63-124. doi: 10.21775/cimb.041.063. Epub 2020 Aug 7. Curr Issues Mol Biol. 2021. PMID: 32764159 Free PMC article. Review.
-
Induction of necroptosis in multinucleated giant cells induced by conditionally replicating syncytial oHSV in co-cultures of cancer cells and non-cancerous cells.Mol Ther Oncol. 2024 Apr 15;32(2):200803. doi: 10.1016/j.omton.2024.200803. eCollection 2024 Jun 20. Mol Ther Oncol. 2024. PMID: 38706990 Free PMC article.
-
Novel mutations in UL24 and gH rescue efficient infection of an HSV vector retargeted to TrkA.Mol Ther Methods Clin Dev. 2023 Jul 3;30:208-220. doi: 10.1016/j.omtm.2023.06.012. eCollection 2023 Sep 14. Mol Ther Methods Clin Dev. 2023. PMID: 37519407 Free PMC article.
-
Potent anti-tumor effects of receptor-retargeted syncytial oncolytic herpes simplex virus.Mol Ther Oncolytics. 2021 Aug 19;22:265-276. doi: 10.1016/j.omto.2021.08.002. eCollection 2021 Sep 24. Mol Ther Oncolytics. 2021. PMID: 34553018 Free PMC article.
-
Cell Fusion and Syncytium Formation in Betaherpesvirus Infection.Viruses. 2021 Sep 30;13(10):1973. doi: 10.3390/v13101973. Viruses. 2021. PMID: 34696402 Free PMC article. Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

