Regulation of GPCR expression through an interaction with CCT7, a subunit of the CCT/TRiC complex
- PMID: 27708139
- PMCID: PMC5170604
- DOI: 10.1091/mbc.E16-04-0224
Regulation of GPCR expression through an interaction with CCT7, a subunit of the CCT/TRiC complex
Abstract
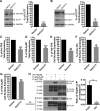
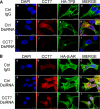
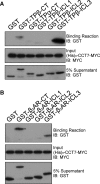
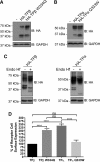
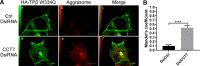
Mechanisms that prevent aggregation and promote folding of nascent G protein-coupled receptors (GPCRs) remain poorly understood. We identified chaperonin containing TCP-1 subunit eta (CCT7) as an interacting partner of the β-isoform of thromboxane A2 receptor (TPβ) by yeast two-hybrid screening. CCT7 coimmunoprecipitated with overexpressed TPβ and β2-adrenergic receptor (β2AR) in HEK 293 cells, but also with endogenous β2AR. CCT7 depletion by small interfering RNA reduced total and cell-surface expression of both receptors and caused redistribution of the receptors to juxtanuclear aggresomes, significantly more so for TPβ than β2AR. Interestingly, Hsp90 coimmunoprecipitated with β2AR but virtually not with TPβ, indicating that nascent GPCRs can adopt alternative folding pathways. In vitro pull-down assays showed that both receptors can interact directly with CCT7 through their third intracellular loops and C-termini. We demonstrate that Trp334 in the TPβ C-terminus is critical for the CCT7 interaction and plays an important role in TPβ maturation and cell-surface expression. Of note, introducing a tryptophan in the corresponding position of the TPα isoform confers the CCT7-binding and maturation properties of TPβ. We show that an interaction with a subunit of the CCT/TCP-1 ring complex (TRiC) chaperonin complex is involved in regulating aggregation of nascent GPCRs and in promoting their proper maturation and expression.
© 2016 Génier et al. This article is distributed by The American Society for Cell Biology under license from the author(s). Two months after publication it is available to the public under an Attribution–Noncommercial–Share Alike 3.0 Unported Creative Commons License (http://creativecommons.org/licenses/by-nc-sa/3.0).
Figures





Similar articles
-
RACK1 regulates the cell surface expression of the G protein-coupled receptor for thromboxane A(2).Traffic. 2008 Mar;9(3):394-407. doi: 10.1111/j.1600-0854.2007.00692.x. Epub 2007 Dec 14. Traffic. 2008. PMID: 18088317
-
WDR36 acts as a scaffold protein tethering a G-protein-coupled receptor, Gαq and phospholipase Cβ in a signalling complex.J Cell Sci. 2011 Oct 1;124(Pt 19):3292-304. doi: 10.1242/jcs.085795. J Cell Sci. 2011. PMID: 21940795
-
Physiological significance of thromboxane A(2) receptor dimerization.J Pharmacol Sci. 2006 Apr;100(4):263-70. doi: 10.1254/jphs.fp0050839. Epub 2006 Mar 25. J Pharmacol Sci. 2006. PMID: 16565578
-
[Regulation of G protein-coupled receptor function by its binding proteins].Yakugaku Zasshi. 2007 Jan;127(1):3-14. doi: 10.1248/yakushi.127.3. Yakugaku Zasshi. 2007. PMID: 17202780 Review. Japanese.
-
Cell signalling through thromboxane A2 receptors.Cell Signal. 2004 May;16(5):521-33. doi: 10.1016/j.cellsig.2003.10.008. Cell Signal. 2004. PMID: 14751539 Review.
Cited by
-
The Function of Different Subunits of the Molecular Chaperone CCT in the Microsporidium Nosema bombycis: NbCCTζ Interacts with NbCCTα.J Fungi (Basel). 2024 Mar 20;10(3):229. doi: 10.3390/jof10030229. J Fungi (Basel). 2024. PMID: 38535237 Free PMC article.
-
Proteomic change by Korean Red Ginseng in the substantia nigra of a Parkinson's disease mouse model.J Ginseng Res. 2018 Oct;42(4):429-435. doi: 10.1016/j.jgr.2017.04.008. Epub 2017 Apr 28. J Ginseng Res. 2018. PMID: 30337802 Free PMC article.
-
Pluripotent stem cell-derived models of neurological diseases reveal early transcriptional heterogeneity.Genome Biol. 2021 Mar 4;22(1):73. doi: 10.1186/s13059-021-02301-6. Genome Biol. 2021. PMID: 33663567 Free PMC article.
-
Sequence grammar underlying the unfolding and phase separation of globular proteins.Mol Cell. 2022 Sep 1;82(17):3193-3208.e8. doi: 10.1016/j.molcel.2022.06.024. Epub 2022 Jul 18. Mol Cell. 2022. PMID: 35853451 Free PMC article.
-
Relax, Cool Down and Scaffold: How to Restore Surface Expression of Folding-Deficient Mutant GPCRs and SLC6 Transporters.Int J Mol Sci. 2017 Nov 14;18(11):2416. doi: 10.3390/ijms18112416. Int J Mol Sci. 2017. PMID: 29135937 Free PMC article. Review.
References
-
- Achour L, Labbé-Jullié C, Scott MGH, Marullo S. An escort for GPCRs: implications for regulation of receptor density at the cell surface. Trends Pharmacol Sci. 2008;29:528–535. - PubMed
-
- Amit M, Weisberg SJ, Nadler-Holly M, McCormack EA, Feldmesser E, Kaganovich D, Willison KR, Horovitz A. Equivalent mutations in the eight subunits of the chaperonin CCT produce dramatically different cellular and gene expression phenotypes. J Mol Biol. 2010;401:532–543. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

