Modeling Ca2+-Bound Troponin in Excitation Contraction Coupling
- PMID: 27708586
- PMCID: PMC5030304
- DOI: 10.3389/fphys.2016.00406
Modeling Ca2+-Bound Troponin in Excitation Contraction Coupling
Abstract
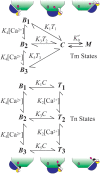
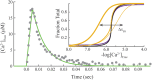
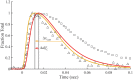
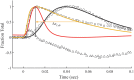
To explain disparate decay rates of cytosolic Ca2+ and structural changes in the thin filaments during a twitch, we model the time course of Ca2+-bound troponin (Tn) resulting from the free Ca2+ transient of fast skeletal muscle. In fibers stretched beyond overlap, the decay of Ca2+ as measured by a change in fluo-3 fluorescence is significantly slower than the intensity decay of the meridional 1/38.5 nm-1 reflection of Tn; this is not simply explained by considering only the Ca2+ binding properties of Tn alone (Matsuo et al., 2010). We apply a comprehensive model that includes the known Ca2+ binding properties of Tn in the context of the thin filament with and without cycling crossbridges. Calculations based on the model predict that the transient of Ca2+-bound Tn correlates with either the fluo-3 time course in muscle with overlapping thin and thick filaments or the intensity of the meridional 1/38.5 nm-1 reflection in overstretched muscle. Hence, cycling crossbridges delay the dissociation of Ca2+ from Tn. Correlation with the fluo-3 fluorescence change is not causal given that the transient of Ca2+-bound Tn depends on sarcomere length, whereas the fluo-3 fluorescence change does not. Transient positions of tropomyosin calculated from the time course of Ca2+-bound Tn are in reasonable agreement with the transient of measured perturbations of the Tn repeat in overlap and non-overlap muscle preparations.
Keywords: EC-coupling; calcium; contraction; excitation; kinetics; model; muscle; troponin.
Figures



Similar articles
-
Structural changes of the regulatory proteins bound to the thin filaments in skeletal muscle contraction by X-ray fiber diffraction.Biochem Biophys Res Commun. 2008 Apr 25;369(1):100-8. doi: 10.1016/j.bbrc.2007.11.088. Epub 2007 Dec 17. Biochem Biophys Res Commun. 2008. PMID: 18082133
-
Regulation of contraction in striated muscle.Physiol Rev. 2000 Apr;80(2):853-924. doi: 10.1152/physrev.2000.80.2.853. Physiol Rev. 2000. PMID: 10747208 Review.
-
Fluorescence resonance energy transfer between residues on troponin and tropomyosin in the reconstituted thin filament: modeling the troponin-tropomyosin complex.J Mol Biol. 2008 Feb 8;376(1):80-91. doi: 10.1016/j.jmb.2007.10.078. Epub 2007 Nov 4. J Mol Biol. 2008. PMID: 18155235
-
Monitoring the structural behavior of troponin and myoplasmic free Ca2+ concentration during twitch of frog skeletal muscle.Biophys J. 2010 Jul 7;99(1):193-200. doi: 10.1016/j.bpj.2010.04.021. Biophys J. 2010. PMID: 20655847 Free PMC article.
-
Sarcomere mechanics in uniform and non-uniform cardiac muscle: a link between pump function and arrhythmias.Prog Biophys Mol Biol. 2008 Jun-Jul;97(2-3):312-31. doi: 10.1016/j.pbiomolbio.2008.02.013. Epub 2008 Feb 15. Prog Biophys Mol Biol. 2008. PMID: 18394686 Review.
Cited by
-
Modeling and simulation of excitation- contraction coupling of fast-twitch skeletal muscle fibers.Technol Health Care. 2020;28(S1):13-24. doi: 10.3233/THC-209003. Technol Health Care. 2020. PMID: 32364140 Free PMC article.
-
Motoneuron-driven computational muscle modelling with motor unit resolution and subject-specific musculoskeletal anatomy.PLoS Comput Biol. 2023 Dec 7;19(12):e1011606. doi: 10.1371/journal.pcbi.1011606. eCollection 2023 Dec. PLoS Comput Biol. 2023. PMID: 38060619 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

