Progressive Dystrophic Pathology in Diaphragm and Impairment of Cardiac Function in FKRP P448L Mutant Mice
- PMID: 27711214
- PMCID: PMC5053477
- DOI: 10.1371/journal.pone.0164187
Progressive Dystrophic Pathology in Diaphragm and Impairment of Cardiac Function in FKRP P448L Mutant Mice
Abstract
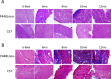
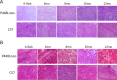
Mutations in the gene for fukutin-related protein represent a subset of muscular dystrophies known as dystroglycanopathies characterized by loss of functionally-glycosylated-alpha-dystroglycan and a wide range of dystrophic phenotypes. Mice generated by our lab containing the P448L mutation in the fukutin-related protein gene demonstrate the dystrophic phenotype similar to that of LGMD2I. Here we examined the morphology of the heart and diaphragm, focusing on pathology of diaphragm and cardiac function of the mutant mice for up to 12 months. Both diaphragm and heart lack clear expression of functionally-glycosylated-alpha-dystroglycan throughout the observed period. The diaphragm undergoes progressive deterioration in histology with increasing amount of centranucleation and inflammation. Large areas of mononuclear cell infiltration and fibrosis of up to 60% of tissue area were detected as early as 6 months of age. Despite a less severe morphology with only patches of mononuclear cell infiltration and fibrosis of ~5% by 12 months of age in the heart, cardiac function is clearly affected. High frequency ultrasound reveals a smaller heart size up to 10 months of age. There are significant increases in myocardial thickness and decrease in cardiac output through 12 months. Dysfunction in the heart represents a key marker for evaluating experimental therapies aimed at cardiac muscle.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



Similar articles
-
Adeno-associated virus-mediated overexpression of LARGE rescues α-dystroglycan function in dystrophic mice with mutations in the fukutin-related protein.Hum Gene Ther Methods. 2014 Jun;25(3):187-96. doi: 10.1089/hgtb.2013.151. Epub 2014 May 2. Hum Gene Ther Methods. 2014. PMID: 24635668 Free PMC article.
-
Ribitol restores functionally glycosylated α-dystroglycan and improves muscle function in dystrophic FKRP-mutant mice.Nat Commun. 2018 Aug 27;9(1):3448. doi: 10.1038/s41467-018-05990-z. Nat Commun. 2018. PMID: 30150693 Free PMC article.
-
Fukutin-related protein is essential for mouse muscle, brain and eye development and mutation recapitulates the wide clinical spectrums of dystroglycanopathies.Hum Mol Genet. 2010 Oct 15;19(20):3995-4006. doi: 10.1093/hmg/ddq314. Epub 2010 Jul 30. Hum Mol Genet. 2010. PMID: 20675713
-
Functions of fukutin, a gene responsible for Fukuyama type congenital muscular dystrophy, in neuromuscular system and other somatic organs.Cent Nerv Syst Agents Med Chem. 2010 Jun 1;10(2):169-79. doi: 10.2174/187152410791196369. Cent Nerv Syst Agents Med Chem. 2010. PMID: 20518731 Review.
-
Muscular Dystrophy with Ribitol-Phosphate Deficiency: A Novel Post-Translational Mechanism in Dystroglycanopathy.J Neuromuscul Dis. 2017;4(4):259-267. doi: 10.3233/JND-170255. J Neuromuscul Dis. 2017. PMID: 29081423 Free PMC article. Review.
Cited by
-
Dose-Dependent Effects of FKRP Gene-Replacement Therapy on Functional Rescue and Longevity in Dystrophic Mice.Mol Ther Methods Clin Dev. 2018 Oct 13;11:106-120. doi: 10.1016/j.omtm.2018.10.004. eCollection 2018 Dec 14. Mol Ther Methods Clin Dev. 2018. PMID: 30417025 Free PMC article.
-
Skeletal, cardiac, and respiratory muscle function and histopathology in the P448Lneo- mouse model of FKRP-deficient muscular dystrophy.Skelet Muscle. 2018 Apr 6;8(1):13. doi: 10.1186/s13395-018-0158-x. Skelet Muscle. 2018. PMID: 29625576 Free PMC article.
-
A limb-girdle muscular dystrophy 2I model of muscular dystrophy identifies corrective drug compounds for dystroglycanopathies.JCI Insight. 2018 Sep 20;3(18):e120493. doi: 10.1172/jci.insight.120493. eCollection 2018 Sep 20. JCI Insight. 2018. PMID: 30232282 Free PMC article.
-
G-quadruplex ligands mediate downregulation of DUX4 expression.Nucleic Acids Res. 2020 May 7;48(8):4179-4194. doi: 10.1093/nar/gkaa146. Nucleic Acids Res. 2020. PMID: 32182342 Free PMC article.
-
Efficacy and muscle safety assessment of fukutin-related protein gene therapy.Mol Ther Methods Clin Dev. 2023 Jun 4;30:65-80. doi: 10.1016/j.omtm.2023.05.022. eCollection 2023 Sep 14. Mol Ther Methods Clin Dev. 2023. PMID: 37361354 Free PMC article.
References
-
- Yoshida M, Ozawa E (1990) Glycoprotein complex anchoring dystrophin to sarcolemma. J Biochem 108: 748–752. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

