Fentanyl for neuropathic pain in adults
- PMID: 27727431
- PMCID: PMC6457928
- DOI: 10.1002/14651858.CD011605.pub2
Fentanyl for neuropathic pain in adults
Abstract
Background: Opioid drugs, including fentanyl, are commonly used to treat neuropathic pain, and are considered effective by some professionals. Most reviews have examined all opioids together. This review sought evidence specifically for fentanyl, at any dose, and by any route of administration. Other opioids are considered in separate reviews.
Objectives: To assess the analgesic efficacy of fentanyl for chronic neuropathic pain in adults, and the adverse events associated with its use in clinical trials.
Search methods: We searched the Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, and Embase from inception to June 2016, together with the reference lists of retrieved articles, and two online study registries.
Selection criteria: We included randomised, double-blind studies of two weeks' duration or longer, comparing fentanyl (in any dose, administered by any route, and in any formulation) with placebo or another active treatment in chronic neuropathic pain.
Data collection and analysis: Two review authors independently searched for studies, extracted efficacy and adverse event data, and examined issues of study quality and potential bias. We did not carry out any pooled analyses. We assessed the quality of the evidence using GRADE.
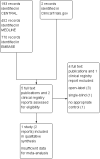
Main results: Only one study met our inclusion criteria. Participants were men and women (mean age 67 years), with postherpetic neuralgia, complex regional pain syndrome, or chronic postoperative pain. They were experiencing inadequate relief from non-opioid analgesics, and had not previously taken opioids for their neuropathic pain. The study used an enriched enrolment randomised withdrawal design. It was adequately blinded, but we judged it at unclear risk of bias for other criteria.Transdermal fentanyl (one-day fentanyl patch) was titrated over 10 to 29 days to establish the maximum tolerated and effective dose (12.5 to 50 µg/h). Participants who achieved a prespecified good level of pain relief with a stable dose of fentanyl, without excessive use of rescue medication or intolerable adverse events ('responders'), were randomised to continue with fentanyl or switch to placebo for 12 weeks, under double-blind conditions. Our prespecified primary outcomes were not appropriate for this study design, but the measures reported do give an indication of the efficacy of fentanyl in this condition.In the titration phase, 1 in 3 participants withdrew because of adverse events or inadequate pain relief, and almost 90% experienced adverse events. Of 258 participants who underwent open-label titration, 163 were 'responders' and entered the randomised withdrawal phase. The number of participants completing the study (and therefore continuing on treatment) without an increase of pain by more than 15/100 was 47/84 (56%) with fentanyl and 28/79 (35%) with placebo. Because only 63% responded sufficiently to enter the randomised withdrawal phase, this implies that only a maximum of 35% of participants entering the study would have had useful pain relief and tolerability with transdermal fentanyl, compared with 22% with placebo. Almost 60% of participants taking fentanyl were 'satisfied' and 'very satisfied' with their treatment at the end of the study, compared with about 40% with placebo. This outcome approximates to our primary outcome of moderate benefit using the Patient Global Impression of Change scale, but the group was enriched for responders and the method of analysis was not clear. The most common adverse events were constipation, nausea, somnolence, and dizziness.There was no information about other types of neuropathic pain, other routes of administration, or comparisons with other treatments.We downgraded the quality of the evidence to very low because there was only one study, with few participants and events, and there was no information about how data from people who withdrew were analysed.
Authors' conclusions: There is insufficient evidence to support or refute the suggestion that fentanyl works in any neuropathic pain condition.
Conflict of interest statement
SD: none known.
CS: none known. She is a specialist pain physician and manages patients with chronic pain.
PC received support from Boston Scientific (2014) for travel and accommodation at a scientific meeting; Boston Scientific does not market drugs. PC is a specialist pain physician and manages patients with chronic pain.
PW: none known.
RK has consulted for Grunenthal Ltd (2014‐15) and MundiPharma Research (2015), and received lecture fees from Grunenthal Ltd (2013‐14) and Pfizer Ltd (2013‐14). He is an Associate Professor in Clinical Pharmacy Practice and Advanced Pharmacy Practitioner.
DA has received lecture fees from Grünenthal (2013, 2014, 2015) and Pfizer (2013, 2016). He is a specialist pain physician and manages patients with chronic pain.
RAM has received grant support from RB relating to individual patient‐level analyses of trial data on ibuprofen in acute pain and the effects of food on drug absorption of analgesics (2013), and from Grünenthal relating to individual patient‐level analyses of trial data regarding tapentadol in osteoarthritis and back pain (2015). He has received honoraria for attending boards with Menarini concerning methods of analgesic trial design (2014), with Novartis about the design of network meta‐analyses (2014), and RB on understanding the pharmacokinetics of drug uptake (2015).
Figures
Update of
References
References to studies included in this review
Arai 2015 {published data only}
-
- Arai T, Kashimoto Y, Ukyo Y, Tominaga Y, Imanaka K. Two placebo‐controlled, randomized withdrawal studies to evaluate the fentanyl 1 day patch in opioid‐naïve patients with chronic pain. Current Medical Research and Opinion 2015;31(12):2207‐18. [DOI: 10.1185/03007995.2015.1092127; JNS020‐JPN‐N02] - DOI - PubMed
-
- Janssen Pharmaceutical KK (Sponsors). A confirmatory study of fentanyl in participants with post‐herpetic neuralgia, complex regional pain syndrome or postoperative pain syndrome. clinicaltrials.gov/ct2/show/record/NCT01008553 (first received 5 November 2009). [CTG: NCT01008553]
References to studies excluded from this review
Canneti 2013 {published data only}
-
- Canneti A, Luzi M, Marco P, Cannata F, Pasqualitto F, Spinoglio A, et al. Safety and efficacy of transdermal buprenorphine and transdermal fentanyl in the treatment of neuropathic pain in AIDS patients. Minerva Anestesiologica 2013;79(8):871‐83. [PUBMED: 23558760] - PubMed
Ding 2014 {published data only}
-
- Ding Y, Yao P, Lan P, Ma J, Wang Z, Hong T. Assessment of transdermal fentanyl combined with gabapentin for malignant neuropathic pain treatment. [Chinese]. Chinese Journal of Clinical Oncology 2014;41:1307‐11. [EMBASE ID: 2014953970]
Kalso 2007 {published data only}
NCT01127100 {published data only}
-
- NCT01127100. Gabapentin versus transdermal fentanyl matrix for chronic neuropathic pain. clinicaltrials.gov/ct2/show/NCT01127100 (first received 19 May 2010). [CTG: NCT01127100]
Additional references
Agarwal 2007
Allan 2001
Allan 2005
-
- Allan L, Richarz U, Simpson K, Slappendel R. Transdermal fentanyl versus sustained release oral morphine in strong‐opioid naïve patients with chronic low back pain. Spine (Phila Pa 1976) 2005;30(22):2484‐90. [PUBMED: 16284584] - PubMed
Baron 2010
Baron 2012
Bouhassira 2008
Bullingham 1984
-
- Bullingham RE, Moore RA, Symonds HW, Allen MC, Baldwin D, McQuay HJ. A novel form of dependency of hepatic extraction ratio of opioids in vivo upon the portal vein concentration of drug: comparison of morphine, diamorphine, fentanyl, methadone and buprenorphine in the chronically cannulated cow. Life Sciences 1984;34(21):2047‐56. - PubMed
Cachia 2011
Dellemijn 1998
Derry 2012
Derry 2013
Derry 2014
Di Franco 2010
-
- Franco M, Iannuccelli C, Atzeni F, Cazzola M, Salaffi F, Valesini G, et al. Pharmacological treatment of fibromyalgia. Clinical and Experimental Rheumatology 2010;28(6 Suppl 63):S110‐6. - PubMed
Dworkin 2008
Finnerup 2015
Franco 2002
-
- Franco ML, Jesus Berro M, Calvo I, Pezonaga L, Sulleiro R, Seoane A, et al. Usefulness of transdermal fentanyl in the management of nonmalignant chronic pain: A prospective, observational, multicenter study. Pain Clinic 2002;14(2):99‐112.
Franklin 2014
Gaskell 2014
Gustorff 2008
Guyatt 2011
Guyatt 2013a
-
- Guyatt G, Oxman AD, Sultan S, Brozek J, Glasziou P, Alonso‐Coello P, et al. GRADE guidelines: 11. Making an overall rating of confidence in effect estimates for a single outcome and for all outcomes. Journal of Clinical Epidemiology 2013;66(2):151‐7. [DOI: 10.1016/j.jclinepi.2012.01.006] - DOI - PubMed
Guyatt 2013b
Hall 2008
Hall 2013
Heiskanen 2009
Higgins 2011
-
- Higgins JPT, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Hoffman 2010
Jadad 1996
Jensen 2011
Kalso 2013
Katusic 1991
Koopman 2009
L'Abbé 1987
-
- L'Abbé KA, Detsky AS, O'Rourke K. Meta‐analysis in clinical research. Annals of Internal Medicine 1987;107:224‐33. - PubMed
Lange 2010
Lunn 2014
Lötsch 2013
McNicol 2013
McQuay 1979
-
- McQuay HJ, Moore RA, Paterson GM, Adams AP. Plasma fentanyl concentrations and clinical observations during and after operation. British Journal of Anaesthesia 1979;51(6):543‐50. - PubMed
McQuay 1998
-
- McQuay H, Moore R. An Evidence‐Based Resource for Pain Relief. Oxford (UK): Oxford University Press, 1998. [ISBN: 0‐19‐263048‐2]
McQuay 2007
-
- McQuay HJ, Smith LA, Moore RA. Chronic pain. In: Stevens A, Raftery J, Mant J, Simpson S editor(s). Health Care Needs Assessment, 3rd Series. Oxford (UK): Radcliffe Publishing, 2007:519‐99. [ISBN: 978‐1‐84619‐063‐6]
Milligan 2001
Mitra 2011
-
- Mitra F, Chowdhury S, Williams G, Shelley M. Comparison of transdermal fentanyl and buprenorphine on clinical outcomes, acceptability and side‐effects on prospective one‐year follow‐up of long‐term persistent pain patients. Australian Pain Society Conference. DArwin, NT Australia: Australian Society of Anaesthetists, 2011; Vol. 39 (4):747‐8.
Moisset 2007
Moore 1998
Moore 2008
-
- Moore RA, Barden J, Derry S, McQuay HJ. Managing potential publication bias. In: McQuay HJ, Kalso E, Moore RA editor(s). Systematic Reviews in Pain Research: Methodology Refined. Seattle (WA): IASP Press, 2008:15‐24. [ISBN: 978‐0‐931092‐69‐5]
Moore 2009
Moore 2010a
Moore 2010b
Moore 2010c
Moore 2010d
Moore 2010e
-
- Moore RA, Moore OA, Derry S, Peloso PM, Gammaitoni AR, Wang H. Responder analysis for pain relief and numbers needed to treat in a meta‐analysis of etoricoxib osteoarthritis trials: bridging a gap between clinical trials and clinical practice. Annals of the Rheumatic Diseases 2010;69(2):374‐9. [DOI: 10.1136/ard.2009.107805] - DOI - PMC - PubMed
Moore 2011a
-
- Moore RA, Straube S, Paine J, Derry S, McQuay HJ. Minimum efficacy criteria for comparisons between treatments using individual patient meta‐analysis of acute pain trials: examples of etoricoxib, paracetamol, ibuprofen, and ibuprofen/paracetamol combinations after third molar extraction. Pain 2011;152(5):982‐9. - PubMed
Moore 2011b
Moore 2012a
Moore 2012b
Moore 2013a
Moore 2013b
Moore 2014a
Moore 2014b
Moore 2014c
Moore 2015
Mystakidou 2003
Nelson 2009
NICE 2013
-
- National Institute for Health and Care Excellence (NICE). Neuropathic pain ‐ pharmacological management: the pharmacological management of neuropathic pain in adults in non‐specialist settings, 2013. www.nice.org.uk/guidance/cg173 (accessed 6 October 2016).
O'Brien 2010
O'Connor 2009
PaPaS 2012
-
- Cochrane Pain, Palliative and Supportive Care Group (PaPaS) author and referee guidance. papas.cochrane.org/papas‐documents (accessed 6 October 2016).
Park 2011
PCA 2015
-
- Prescribing and Medicines Team, Health and Social Care Information Centre. Prescription Cost Analysis, England 2014. content.digital.nhs.uk/catalogue/PUB17274/pres‐cost‐anal‐eng‐2014‐rep.pdf (accessed 6 October 2016):119‐21. [ISBN: 978‐1‐78386‐365‐5]
Rappaport 1994
-
- Rappaport ZH, Devor M. Trigeminal neuralgia: the role of self‐sustaining discharge in the trigeminal ganglion. Pain 1994;56:127‐38. - PubMed
Raptis 2014
RevMan 2014 [Computer program]
-
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Soni 2013
Straube 2008
-
- Straube S, Derry S, McQuay HJ, Moore RA. Enriched enrolment: definition and effects of enrichment and dose in trials of pregabalin and gabapentin in neuropathic pain. A systematic review. British Journal of Clinical Pharmacology 2008;66(2):266‐75. [DOI: 10.1111/j.1365-2125.2008.03200] - DOI - PMC - PubMed
Straube 2010
Sultan 2008
Torrance 2006
Tracey 2011
Treede 2008
Trescot 2008
-
- Trescot AM, Datta S, Lee M, Hansen H. Opioid pharmacology. Pain Physician 2008;11:S133‐53. - PubMed
van Hecke 2014
Vo 2009
von Hehn 2012
Vos 2012
-
- Vos T, Flaxman AD, Naghavi M, Lozano R, Michaud C, Ezzati M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990‐2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012;380(9859):2163‐96. [DOI: 10.1016/S0140-6736(12)61729-2] - DOI - PMC - PubMed
Weisberg 2014
Wiffen 2013
Wiffen 2015
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous