Alemtuzumab improves preexisting disability in active relapsing-remitting MS patients
- PMID: 27733571
- PMCID: PMC5109953
- DOI: 10.1212/WNL.0000000000003319
Alemtuzumab improves preexisting disability in active relapsing-remitting MS patients
Abstract
Objective: To characterize effects of alemtuzumab treatment on measures of disability improvement in patients with relapsing-remitting multiple sclerosis (RRMS) with inadequate response (≥1 relapse) to prior therapy.
Methods: Comparison of Alemtuzumab and Rebif Efficacy in Multiple Sclerosis (CARE-MS) II, a 2-year randomized, rater-blinded, active-controlled, head-to-head, phase 3 trial, compared efficacy and safety of alemtuzumab 12 mg with subcutaneous interferon-β-1a (SC IFN-β-1a) 44 μg in patients with RRMS. Prespecified and post hoc disability outcomes based on Expanded Disability Status Scale (EDSS), Multiple Sclerosis Functional Composite (MSFC), and Sloan low-contrast letter acuity (SLCLA) are reported, focusing on improvement of preexisting disability in addition to slowing of disability accumulation.
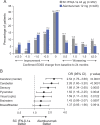
Results: Alemtuzumab-treated patients were more likely than SC IFN-β-1a-treated patients to show improvement in EDSS scores (p < 0.0001) on all 7 functional systems. Significantly more alemtuzumab patients demonstrated 6-month confirmed disability improvement. The likelihood of improved vs stable/worsening MSFC scores was greater with alemtuzumab than SC IFN-β-1a (p = 0.0300); improvement in MSFC scores with alemtuzumab was primarily driven by the upper limb coordination and dexterity domain. Alemtuzumab-treated patients had more favorable changes from baseline in SLCLA (2.5% contrast) scores (p = 0.0014) and MSFC + SLCLA composite scores (p = 0.0097) than SC IFN-β-1a-treated patients.
Conclusions: In patients with RRMS and inadequate response to prior disease-modifying therapies, alemtuzumab provides greater benefits than SC IFN-β-1a across several disability outcomes, reflecting improvement of preexisting disabilities.
Classification of evidence: This study provides Class I evidence (based on rater blinding and a balance in baseline characteristics between arms) that alemtuzumab modifies disability measures favorably compared with SC IFN-β-1a.
© 2016 American Academy of Neurology.
Figures


Comment in
-
Sustained reduction of MS disability: New player in comparing disease-modifying treatments.Neurology. 2016 Nov 8;87(19):1966-1967. doi: 10.1212/WNL.0000000000003314. Epub 2016 Oct 12. Neurology. 2016. PMID: 27733564 No abstract available.
References
-
- Gold R, Kappos L, Arnold DL, et al. Placebo-controlled phase 3 study of oral BG-12 for relapsing multiple sclerosis. N Engl J Med 2012;367:1098–1107. - PubMed
-
- Johnson KP, Brooks BR, Cohen JA, et al. Copolymer 1 reduces relapse rate and improves disability in relapsing-remitting multiple sclerosis: results of a phase III multicenter, double-blind placebo-controlled trial: The Copolymer 1 Multiple Sclerosis Study Group. Neurology 1995;45:1268–1276. - PubMed
-
- Kappos L, Radue EW, O'Connor P, et al. A placebo-controlled trial of oral fingolimod in relapsing multiple sclerosis. N Engl J Med 2010;362:387–401. - PubMed
-
- O'Connor P, Wolinsky JS, Confavreux C, et al. Randomized trial of oral teriflunomide for relapsing multiple sclerosis. N Engl J Med 2011;365:1293–1303. - PubMed
-
- Polman CH, O'Connor PW, Havrdova E, et al. A randomized, placebo-controlled trial of natalizumab for relapsing multiple sclerosis. N Engl J Med 2006;354:899–910. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
