Empagliflozin decreases myocardial cytoplasmic Na+ through inhibition of the cardiac Na+/H+ exchanger in rats and rabbits
- PMID: 27752710
- PMCID: PMC6518059
- DOI: 10.1007/s00125-016-4134-x
Empagliflozin decreases myocardial cytoplasmic Na+ through inhibition of the cardiac Na+/H+ exchanger in rats and rabbits
Abstract
Aims/hypothesis: Empagliflozin (EMPA), an inhibitor of the renal sodium-glucose cotransporter (SGLT) 2, reduces the risk of cardiovascular death in patients with type 2 diabetes. The underlying mechanism of this effect is unknown. Elevated cardiac cytoplasmic Na+ ([Na+]c) and Ca2+ ([Ca2+]c) concentrations and decreased mitochondrial Ca2+ concentration ([Ca2+]m) are drivers of heart failure and cardiac death. We therefore hypothesised that EMPA would directly modify [Na+]c, [Ca2+]c and [Ca2+]m in cardiomyocytes.
Methods: [Na+]c, [Ca2+]c, [Ca 2+]m and Na+/H+ exchanger (NHE) activity were measured fluorometrically in isolated ventricular myocytes from rabbits and rats.
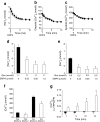
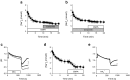
Results: An increase in extracellular glucose, from 5.5 mmol/l to 11 mmol/l, resulted in increased [Na+]c and [Ca2+]c levels. EMPA treatment directly inhibited NHE flux, caused a reduction in [Na+]c and [Ca2+]c and increased [Ca2+]m. After pretreatment with the NHE inhibitor, Cariporide, these effects of EMPA were strongly reduced. EMPA also affected [Na+]c and NHE flux in the absence of extracellular glucose.
Conclusions/interpretation: The glucose lowering kidney-targeted agent, EMPA, demonstrates direct cardiac effects by lowering myocardial [Na+]c and [Ca2+]c and enhancing [Ca2+]m, through impairment of myocardial NHE flux, independent of SGLT2 activity.
Keywords: Calcium; Cardiac death; Diabetes; Glucose; Heart failure; Sodium.
Conflict of interest statement
Data availability
All data is available from the authors upon request.
Duality of interest
The authors declare that there is no duality of interest associated with this manuscript.
Contribution statement
AB, CAS, RCIW, and GJMS contributed to the conception and design, planning of the analysis, and acquisition and interpretation of data, and reviewed and edited the manuscript. JWTF contributed to the analysis planning and interpretation of data, and reviewed and edited the manuscript. RC and CJZ contributed to the conception and design, analysis and interpretation of data, drafting and editing of the manuscript. All authors were fully responsible for all content and editorial decisions, and approved the final version. CJZ is the guarantor of this work.
Figures
References
-
- Marx N, McGuire DK. Sodium-glucose cotransporter-2 inhibitor for the reduction of cardiovascular events in high-risk patients with diabetes mellitus. Eur Heart J. 2016 - PubMed
-
- Baartscheer A, Schumacher CA, Borren MM, Belterman CN, Coronel R, Fiolet JW. Increased Na+/H+-exchange activity is the cause of increased [Na+]i and underlies disturbed calcium handling in the rabbit pressure and volume overload heart failure model. Cardiovasc Res. 2003;57:1015–1024. doi: 10.1016/S0008-6363(02)00809-X. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

