CD40L is transferred to antigen-presenting B cells during delivery of T-cell help
- PMID: 27753080
- PMCID: PMC5233572
- DOI: 10.1002/eji.201646504
CD40L is transferred to antigen-presenting B cells during delivery of T-cell help
Abstract
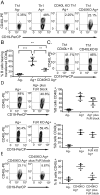
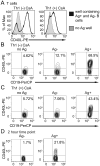
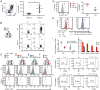
The delivery of T-cell help to B cells is antigen-specific, MHC-restricted, and CD40L (CD154) dependent. It has been thought that when a T cell recognizes an antigen-presenting B cell, CD40L expressed on the T-cell surface engages with CD40 on the surface of B cells as long as the cells remain conjugated. By adding fluorescently labeled anti-CD40L antibody during overnight incubation of antigen-presenting B cells with antigen-specific T cells, we discovered that CD40L does not remain on the surface of the T cell, but it is transferred to and endocytosed by B cells receiving T-cell help. In the presence of anti-CD40L antibody, transferred CD40L is nearly absent on bystander B cells that are not presenting antigen, and the bystander cells do not become activated. Because transfer of CD40L to B cells correlates with B-cell activation, we speculate that persistence of helper T-cell-derived CD40L on or in B cells could permit sustained CD40 signaling enabling survival and proliferation of antigen-presenting B cells following brief interactions with helper T cells in vivo in germinal centers.
Keywords: CD154; CD40 ligand; CD40L; Immunological synapse; Membrane transfer; T-cell help.
© 2016 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures


Comment in
-
Help to go: T cells transfer CD40L to antigen-presenting B cells.Eur J Immunol. 2017 Jan;47(1):31-34. doi: 10.1002/eji.201646786. Epub 2016 Dec 22. Eur J Immunol. 2017. PMID: 28004382
Similar articles
-
Despite disorganized synapse structure, Th2 cells maintain directional delivery of CD40L to antigen-presenting B cells.PLoS One. 2017 Oct 12;12(10):e0186573. doi: 10.1371/journal.pone.0186573. eCollection 2017. PLoS One. 2017. PMID: 29023539 Free PMC article.
-
Immunological synapse formation licenses CD40-CD40L accumulations at T-APC contact sites.J Immunol. 2004 Sep 15;173(6):3647-52. doi: 10.4049/jimmunol.173.6.3647. J Immunol. 2004. PMID: 15356109
-
Help to go: T cells transfer CD40L to antigen-presenting B cells.Eur J Immunol. 2017 Jan;47(1):31-34. doi: 10.1002/eji.201646786. Epub 2016 Dec 22. Eur J Immunol. 2017. PMID: 28004382
-
Functions of CD40 and Its Ligand, gp39 (CD40L).Crit Rev Immunol. 2017;37(2-6):371-420. doi: 10.1615/CritRevImmunol.v37.i2-6.100. Crit Rev Immunol. 2017. PMID: 29773027 Review.
-
Molecular mechanism and function of CD40/CD40L engagement in the immune system.Immunol Rev. 2009 May;229(1):152-72. doi: 10.1111/j.1600-065X.2009.00782.x. Immunol Rev. 2009. PMID: 19426221 Free PMC article. Review.
Cited by
-
The Biological Significance of Trogocytosis.Results Probl Cell Differ. 2024;73:87-129. doi: 10.1007/978-3-031-62036-2_5. Results Probl Cell Differ. 2024. PMID: 39242376 Free PMC article.
-
Germinal center expansion but not plasmablast differentiation is proportional to peptide-MHCII density via CD40-CD40L signaling strength.Cell Rep. 2022 May 3;39(5):110763. doi: 10.1016/j.celrep.2022.110763. Cell Rep. 2022. PMID: 35508132 Free PMC article.
-
CD4+ T Cell-Released Extracellular Vesicles Potentiate the Efficacy of the HBsAg Vaccine by Enhancing B Cell Responses.Adv Sci (Weinh). 2019 Sep 30;6(23):1802219. doi: 10.1002/advs.201802219. eCollection 2019 Dec. Adv Sci (Weinh). 2019. PMID: 31832305 Free PMC article.
-
B cell peripheral tolerance is promoted by cathepsin B protease.Proc Natl Acad Sci U S A. 2023 Apr 18;120(16):e2300099120. doi: 10.1073/pnas.2300099120. Epub 2023 Apr 11. Proc Natl Acad Sci U S A. 2023. PMID: 37040412 Free PMC article.
-
B Cell Endosomal RAB7 Promotes TRAF6 K63 Polyubiquitination and NF-κB Activation for Antibody Class-Switching.J Immunol. 2020 Mar 1;204(5):1146-1157. doi: 10.4049/jimmunol.1901170. Epub 2020 Jan 13. J Immunol. 2020. PMID: 31932498 Free PMC article.
References
-
- Noelle RJ, Ledbetter JA, Aruffo A. Immunol Today. 1992;13:431–433. - PubMed
-
- Schoenberger SP, Toes RE, van der Voort EI, Offringa R, Melief CJ. Nature. 1998;393:480–483. - PubMed
-
- Grewal IS, Flavell RA. Annu Rev Immunol. 1998;16:111–135. - PubMed
-
- Parker DC. Annu Rev Immunol. 1993;11:331–360. - PubMed
-
- Han S, Hathcock K, Zheng B, Kepler TB, Hodes R, Kelsoe G. J Immunol. 1995;155:556–567. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

