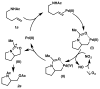
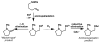Palladium-Catalyzed Aerobic Intramolecular Aminoacetoxylation of Alkenes Enabled by Catalytic Nitrate
- PMID: 27754689
- PMCID: PMC5512709
- DOI: 10.1021/acs.orglett.6b02722
Palladium-Catalyzed Aerobic Intramolecular Aminoacetoxylation of Alkenes Enabled by Catalytic Nitrate
Abstract
A mild aerobic intramolecular aminoacetoxylation method for the synthesis of pyrrolidine and indoline derivatives was achieved using molecular oxygen as the oxidant. A catalytic NOx species acts as an electron transfer mediator to access a high-valent palladium intermediate as the presumed active oxidant.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Similar articles
-
Intramolecular aminocyanation of alkenes by cooperative palladium/boron catalysis.J Am Chem Soc. 2014 Mar 12;136(10):3732-5. doi: 10.1021/ja4122632. Epub 2014 Mar 3. J Am Chem Soc. 2014. PMID: 24580141
-
Palladium(II)-catalyzed enantio- and diastereoselective synthesis of pyrrolidine derivatives.Org Lett. 2012 Aug 17;14(16):4074-7. doi: 10.1021/ol3016989. Epub 2012 Aug 8. Org Lett. 2012. PMID: 22873944 Free PMC article.
-
Novel palladium-catalyzed acyloxylation/cyclization of 2-(3'-alkenyl)indoles.Org Lett. 2009 Jun 4;11(11):2381-4. doi: 10.1021/ol9007348. Org Lett. 2009. PMID: 19419163
-
Pd(0)-catalyzed oxy- and aminoalkynylation of olefins for the synthesis of tetrahydrofurans and pyrrolidines.Org Lett. 2011 Dec 2;13(23):6324-7. doi: 10.1021/ol2029383. Epub 2011 Nov 9. Org Lett. 2011. PMID: 22070183
-
Enantioselective Dicarbofunctionalization of Unactivated Alkenes by Palladium-Catalyzed Tandem Heck/Suzuki Coupling Reaction.Angew Chem Int Ed Engl. 2019 Oct 7;58(41):14653-14659. doi: 10.1002/anie.201907840. Epub 2019 Sep 5. Angew Chem Int Ed Engl. 2019. PMID: 31420928 Review.
Cited by
-
Directed, Palladium(II)-Catalyzed Intermolecular Aminohydroxylation of Alkenes Using a Mild Oxidation System.Org Lett. 2018 Jul 6;20(13):3853-3857. doi: 10.1021/acs.orglett.8b01440. Epub 2018 Jun 11. Org Lett. 2018. PMID: 29888604 Free PMC article.
-
Palladium and copper co-catalyzed chloro-arylation of gem-difluorostyrenes - use of a nitrite additive to suppress β-F elimination.Chem Sci. 2024 Oct 1;15(42):17571-8. doi: 10.1039/d4sc04939j. Online ahead of print. Chem Sci. 2024. PMID: 39386912 Free PMC article.
-
Preparation of Key Intermediates for the Syntheses of Coenzyme Q10 and Derivatives by Cross-Metathesis Reactions.Molecules. 2020 Jan 21;25(3):448. doi: 10.3390/molecules25030448. Molecules. 2020. PMID: 31973220 Free PMC article.
References
-
-
Selected reviews for Pd-catalyzed alkene difunctionalization reactions: Muñiz K. Angew Chem Int Ed Engl. 2009;48:9412–9423.Xu LM, Li BJ, Yang Z, Shi ZJ. Chem Soc Rev. 2010;39:712–733.Sehnal P, Taylor RJK, Fairlamb IJS. Chem Rev. 2010;110:824–889.Hickman AJ, San-ford MS. Nature. 2012;484:177–185.Coombs JR, Morken JP. Angew Chem Int Ed. 2016;55:2636–2649.
-
-
-
For Pd-catalyzed amination reactions: McDonald RI, Liu G, Stahl SS. Chem Rev. 2011;111:2981–3019.Liu G, Stahl SS. J Am Chem Soc. 2007;129:6328–6335.Bertrand MB, Neukom JD, Wolfe JP. J Org Chem. 2008;73:8851–8860.Muñiz K, Hövelmann CH, Streuff J. J Am Chem Soc. 2008;130:763–773.Ye X, Liu G, Popp BV, Stahl SS. J Org Chem. 2011;76:1031–1044.
-
-
-
Selected examples of alkene difunctionalization using high-valent iodine reagents: Alexanian EJ, Lee C, Sorensen EJ. J Am Chem Soc. 2005;127:7690–7691.Streuff J, Hövelmann CH, Nieger M, Muñiz K. J Am Chem Soc. 2005;127:14586–14587.Liu G, Stahl SS. J Am Chem Soc. 2006;128:7179–7181.Desai LV, Sanford MS. Angew Chem Int Ed Engl. 2007;46:5737–5740.Neufeldt SR, Sanford MS. Org Lett. 2013;15:46–49.Martínez C, Wu Y, Weinstein AB, Stahl SS, Liu G, Muñiz K. J Org Chem. 2013;78:6309–6315.Chen H, Kaga A, Chiba S. Org Lett. 2014;16:6136–6139.
-
-
-
For alkene difunctionalization using NFSI as oxidant: Sib-bald PA, Rosewall CF, Swartz RD, Michael FE. J Am Chem Soc. 2009;131:15945–15951.Liskin DV, Sibbald PA, Rosewall CF, Michael FE. J Org Chem. 2010;75:6294–6296.Engle KM, Mei TS, Wang X, Yu JQ. Angew Chem Int Ed. 2011;50:1478–1491.Ingalls EL, Sibbald PA, Kaminsky W, Michael FE. J Am Chem Soc. 2013;135:8854–8856.
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources