Single Channel Recordings Reveal Differential β2 Subunit Modulations Between Mammalian and Drosophila BKCa(β2) Channels
- PMID: 27755549
- PMCID: PMC5068790
- DOI: 10.1371/journal.pone.0163308
Single Channel Recordings Reveal Differential β2 Subunit Modulations Between Mammalian and Drosophila BKCa(β2) Channels
Abstract
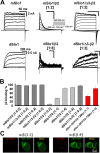
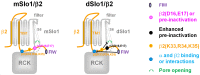
Large-conductance Ca2+- and voltage-activated potassium (BK) channels are widely expressed in tissues. As a voltage and calcium sensor, BK channels play significant roles in regulating the action potential frequency, neurotransmitter release, and smooth muscle contraction. After associating with the auxiliary β2 subunit, mammalian BK(β2) channels (mouse or human Slo1/β2) exhibit enhanced activation and complete inactivation. However, how the β2 subunit modulates the Drosophila Slo1 channel remains elusive. In this study, by comparing the different functional effects on heterogeneous BK(β2) channel, we found that Drosophila Slo1/β2 channel exhibits "paralyzed"-like and incomplete inactivation as well as slow activation. Further, we determined three different modulations between mammalian and Drosophila BK(β2) channels: 1) dSlo1/β2 doesn't have complete inactivation. 2) β2(K33,R34,K35) delays the dSlo1/Δ3-β2 channel activation. 3) dSlo1/β2 channel has enhanced pre-inactivation than mSlo1/β2 channel. The results in our study provide insights into the different modulations of β2 subunit between mammalian and Drosophila Slo1/β2 channels and structural basis underlie the activation and pre-inactivation of other BK(β) complexes.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



Similar articles
-
Modulation of BK channel gating by the ß2 subunit involves both membrane-spanning and cytoplasmic domains of Slo1.J Neurosci. 2010 Dec 1;30(48):16170-9. doi: 10.1523/JNEUROSCI.2323-10.2010. J Neurosci. 2010. PMID: 21123563 Free PMC article.
-
Mechanism of the modulation of BK potassium channel complexes with different auxiliary subunit compositions by the omega-3 fatty acid DHA.Proc Natl Acad Sci U S A. 2013 Mar 19;110(12):4822-7. doi: 10.1073/pnas.1222003110. Epub 2013 Mar 4. Proc Natl Acad Sci U S A. 2013. PMID: 23487786 Free PMC article.
-
A point mutation in the human Slo1 channel that impairs its sensitivity to omega-3 docosahexaenoic acid.J Gen Physiol. 2013 Nov;142(5):507-22. doi: 10.1085/jgp.201311061. Epub 2013 Oct 14. J Gen Physiol. 2013. PMID: 24127525 Free PMC article.
-
A BK (Slo1) channel journey from molecule to physiology.Channels (Austin). 2013 Nov-Dec;7(6):442-58. doi: 10.4161/chan.26242. Epub 2013 Sep 11. Channels (Austin). 2013. PMID: 24025517 Free PMC article. Review.
-
Protein Network Interacting with BK Channels.Int Rev Neurobiol. 2016;128:127-61. doi: 10.1016/bs.irn.2016.03.003. Epub 2016 Mar 28. Int Rev Neurobiol. 2016. PMID: 27238263 Review.
Cited by
-
Rectification ratio based determination of disulfide bonds of β2 extracellular loop of BK channel.Channels (Austin). 2019 Dec;13(1):17-32. doi: 10.1080/19336950.2018.1551660. Channels (Austin). 2019. PMID: 30477399 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

