Malignant Pleural Effusion and ascites Induce Epithelial-Mesenchymal Transition and Cancer Stem-like Cell Properties via the Vascular Endothelial Growth Factor (VEGF)/Phosphatidylinositol 3-Kinase (PI3K)/Akt/Mechanistic Target of Rapamycin (mTOR) Pathway
- PMID: 27756837
- PMCID: PMC5207183
- DOI: 10.1074/jbc.M116.753236
Malignant Pleural Effusion and ascites Induce Epithelial-Mesenchymal Transition and Cancer Stem-like Cell Properties via the Vascular Endothelial Growth Factor (VEGF)/Phosphatidylinositol 3-Kinase (PI3K)/Akt/Mechanistic Target of Rapamycin (mTOR) Pathway
Abstract
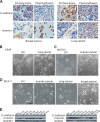
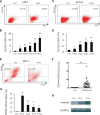
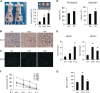
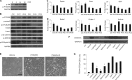
Malignant pleural effusion (PE) and ascites, common clinical manifestations in advanced cancer patients, are associated with a poor prognosis. However, the biological characteristics of malignant PE and ascites are not clarified. Here we report that malignant PE and ascites can induce a frequent epithelial-mesenchymal transition program and endow tumor cells with stem cell properties with high efficiency, which promotes tumor growth, chemoresistance, and immune evasion. We determine that this epithelial-mesenchymal transition process is mainly dependent on VEGF, one initiator of the PI3K/Akt/mechanistic target of rapamycin (mTOR) pathway. From the clinical observation, we define a therapeutic option with VEGF antibody for malignant PE and ascites. Taken together, our findings clarify a novel biological characteristic of malignant PE and ascites in cancer progression and provide a promising and available strategy for cancer patients with recurrent/refractory malignant PE and ascites.
Keywords: VEGF; ascites; cancer biology; cancer stem cells; epithelial-mesenchymal transition (EMT); malignant pleural effusion; tumor therapy.
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures
References
-
- Runyon B. A. (1994) Care of patients with ascites. N. Engl. J. Med. 330, 337–342 - PubMed
-
- Shen-Gunther J., and Mannel R. S. (2002) Ascites as a predictor of ovarian malignancy. Gynecol. Oncol. 87, 77–83 - PubMed
-
- Ryu J. S., Ryu H. J., Lee S. N., Memon A., Lee S. K., Nam H. S., Kim H. J., Lee K. H., Cho J. H., and Hwang S. S. (2014) Prognostic impact of minimal pleural effusion in non-small-cell lung cancer. J. Clin. Oncol. 32, 960–967 - PubMed
-
- Tan D. S., Agarwal R., and Kaye S. B. (2006) Mechanisms of transcoelomic metastasis in ovarian cancer. Lancet Oncol. 7, 925–934 - PubMed
-
- Kolomainen D. F., A'Hern R., Coxon F. Y., Fisher C., King D. M., Blake P. R., Barton D. P., Shepherd J. H., Kaye S. B., and Gore M. E. (2003) Can patients with relapsed, previously untreated, stage I epithelial ovarian cancer be successfully treated with salvage therapy? J. Clin. Oncol. 21, 3113–3118 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

